Motor vehicle travel and driving has become a major component of daily living in the developed world. Sudden incapacity occurring while driving can result in accidents that may be fatal both for the driver and for bystanders. The regulatory approach aims to balance the risk to bystanders against the highly valued individual freedom of motorised mobility. In this article we review some of the concepts that are pivotal to this balancing act, compare variations in national approaches to regulation and ask whether it is time for some of the accepted tenets to be revised.
Syncope and Incapacitation as a Result of Arrhythmia
Sudden incapacity can result from a syncopal event, a sudden cardiac death (SCD), or a neurological event such as seizure or stroke. Syncope is defined as a transient loss of consciousness event that results from general brain hypoperfusion.1 Syncope can be neurally mediated, caused by orthostatic hypotension or by cardiac conditions – mostly arrhythmic events (either brady- or tachyarrhythmias). SCD is an unexpected death from a cardiac cause, which occurs within one hour from the start of any cardiac-related symptoms. It is irreversible if prompt resuscitation is not applied. SCD is mostly arrhythmic in nature, with ventricular tachycardia (VT) and VF responsible for >75% of cases.2
Cardiac pacemakers are used to treat patients with – or those at risk of developing – significant bradyarrhythmias, while ICDs are used to treat patients who are at high risk of SCD as a result of VT/VF. While pacemakers can effectively prevent the occurrence of bradyarrhythmias, ICDs do not prevent VT/VF but treat those rhythms once they happen by either overdrive antitachycardia pacing (ATP) or internal cardioversion. Syncope may still occur in patients who develop VT/VF despite having an ICD because of the time delay between arrhythmia occurrence, effective treatment and restoration of normal brain perfusion.
The following discussion will focus on the driving restrictions in patients at risk of syncope and cardiac arrhythmias associated with sudden incapacity.
Risk Estimate of Motor Vehicle Accident Fatalities in Patients at Risk of Syncope and Cardiac Arrhythmias
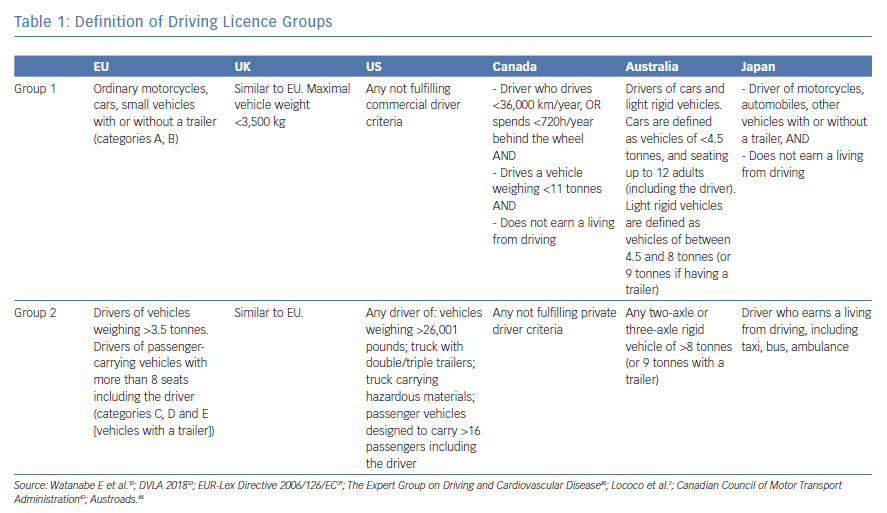
Driving Licence Categories
Driving licences are generally divided into private (group 1), and commercial (group 2). The definition of private and commercial drivers varies somewhat between countries (Table 1) but, in general, a private driver is a licensed driver who does not earn a living from driving and a commercial driver is a driver who earns a living from driving and/or is licensed to drive large passenger or goods-carrying vehicles. Categories for taxi drivers vary between group 1 and group 2 standards and may be locally determined.
General Statistics on Road Traffic Accidents
Driving a car is a central part of life in developed societies. For example, more than 85% of Americans own a car and almost 270 million vehicles are registered in the US alone (of which more than 190 million are “light duty, short wheel base” vehicles).3 However, motor vehicle accidents are a leading cause of death worldwide. Road injuries – including accidents involving all forms of road transportation systems, and pedestrians – killed more than 1.4 million people in 2016.4 In UK, 1,793 people were killed in road accidents in 2017, 44% of which were drivers.5
The risk of death related to driving is highly variable between countries. Road traffic death rates in low- and middle-income countries are more than double those in high-income countries.6 However, wide disparities exist even among developed countries. For example, per capita road fatalities in the US are almost double those in Denmark, and in 2016 there was almost a fourfold difference in road accident fatalities between the ‘safest’ and the ‘least safe’ European countries: Norway (26 fatalities per million population) and Romania (97 fatalities per million population).7,8 While some of the difference may relate to mileage driven, cultural approaches to risk are also an important factor.
Accident and fatality rates also vary according to age. The rate of death in car accidents is highest in individuals aged 20–29 years and those older than 80 years – 16.0 per million population in the UK – and lowest in the middle-aged population – 6.8 per million population in the UK – in those aged 40–49 years.9 In young adults aged 20–29, road traffic accidents are the leading cause of death worldwide.6 Elderly people are at risk of road traffic accidents for a variety of reasons, such as a slower reaction time, depth perception change, vision and hearing problems, decreased ability to focus and medical problems. Compared with younger individuals – for who speed is a major cause of road accidents while driving – physiological and perceptual decline is the major cause of road accidents while driving in older individuals.10
Acceptable Risk of Road Traffic Accidents
Driving carries risk but is a major part of life in many societies, so it follows that these societies accept an intrinsic risk of harm (RH) to self and others because of driving. Nationally defined regulations have implicitly balanced risk and benefit for decades. An attempt to formalise this balancing act emerged from a Canadian Cardiovascular Society conference in 1992 (updated in 2003).11 In this document, the annual RH as a result of driving was defined as:
RH = TD × V × SCI × Ac
where:
- TD is the time spent driving;
- V is the type of vehicle;
- SCI is the risk of sudden incapacitation; and
- Ac is the probability that an episode of sudden incapacitation will result in a fatal or injury-producing accident.
- TD is 0.25 (25%) for professional drivers because the average time spent driving is 6 hours per day; and 0.04 (4%) for social drivers because they spend, on average, 1 hour driving per day.
- V is 1 for trucks and 0.28 for family cars because, on average, accidents involving trucks cause 7.2% of fatalities, despite causing only 2.0% of road accidents (2.0 ÷ 7.2% = 0.28).
- SCI is 0.01 (1%), which was the estimated annual risk of SCD of a truck driver who had not had an acute MI within the previous 3 months, is in functional class I (asymptomatic), has a negative exercise tolerance test, is able to perform at least seven metabolic equivalents of task during the treadmill test, and has no documented ventricular arrhythmias. This driver was historically allowed to drive by Canadian laws, so this was set as the acceptable risk threshold in the RH formula. The 1% mortality per year also holds true for men in the Western population aged >65 years and this limit has been used for maximal annual risk allowance for commercial pilots in aviation risk assessment (the 1% rule).12
- Ac is 0.02 (2%) because only 2% of accidents caused by drivers suffering SCD or sudden incapacity while driving has resulted in harm or death of other road traffic users or bystanders.
Using these assumptions, the annual risk of death or injury to others from allowing a truck driver to drive is approximately 1 in 20,000 (0.005%). It can be estimated that a private driver has a 22-times lower RH compared with a professional driver (TDprofessional ÷ TDsocial × Vtruck ÷ Vcar= 0.25 ÷ 0.04 × 1.00 ÷ 0.28). In other words, a private driver with a SCI of 22% has the same RH as a professional driver with a SCI of 1%.11
There are some important caveats associated with the RH formula. Importantly, TD and V were calculated based on the Ontario Road Safety Annual Report from 1987. The Ac was estimated from reports published between 1974 and 1990. However, road traffic accident rates have declined over the years in high-income countries, but increased in low-income countries.6 In addition, the safety (for drivers, passengers and pedestrians) of new cars sold by respected manufacturers has dramatically increased in the last 20 years. For example, in 1997, all but one of the 20 cars tested by the European New Car Assessment Programme received less than 3/5 stars at crash-tests and the one exception received 4/5 stars, while in 2017 – under far more stringent crash-test safety assessment criteria – 44 of 70 (63%) of the tested cars received the maximum 5/5 stars.13 The actual RH may now be less than the 0.005% calculated in 1992. An assumption in the Canadian model is that all groups of drivers share the same maximum RH threshold, but this ignores the possibility that society would place a different value on different types of driving, for example delivering food or medical supplies versus private driving for pleasure or leisure.
Ideally, societally acceptable RH should be calculated at least nationally, based on specific country road accident profiles, type of roads, types of vehicles, age and gender, individual times spent driving, and so on. In future, it is conceivable that individual medical risk and driving behaviour could be tested against a societal RH threshold using knowledge of an individual patient’s medical condition and a black box device to monitor distance travelled and driving behaviour.
Risk Assessment for Patients at Risk of Syncope and Cardiac Arrhythmias Associated with Sudden Incapacity
Syncope
Data from the Framingham Heart Study suggest that the incidence of syncope in the general population is between 3% and 6% at 10 years.14 Among patients with syncope, 3–10% of syncopal events appear while driving; 85% of these patients have recurrent syncope but in a few the first syncope occurs while driving.15 The causes of syncope while driving are the same as for the general population; 35–38% are neurally mediated, 5–7% are caused equally by orthostatic hypotension, bradyarrhythmias and VTs, and 2–4% are caused by supraventricular tachycardias (almost all of them either atrioventricular nodal re-entrant tachycardia [AVNRT] or atrioventricular re-entrant tachycardia [AVRT]).16 During long-term follow up (8 years), the recurrence rate of syncope was similar in patients who had experienced the first syncopal event while driving and those who had not; 34–39% for neurally-mediated syncope, 7–13% for bradyarrhythmias, and 3–4% for VT and supraventricular tachycardias. Malignant arrhythmias causing SCD (fast VT/VF) are rare. A retrospective study performed in Germany estimated that 0.4% of all road traffic accidents are caused by the driver having a SCD event while driving.17
Overall, patients with a history of syncope have a higher risk of motor vehicle accidents compared with asymptomatic subjects. A Danish nationwide survey identified more than 41,000 patients with syncope and compared their motor vehicle accident rate to the general Danish population.18 During an average 2-year follow up, 4.4% of patients had a vehicle accident, 23.7% of which led to major injury and 0.3% to death. When an accident occurred there was no difference in the risk of serious injury between the syncope and general populations. The crude incidence rate of motor vehicle accident was 1.83-times higher in patients with syncope compared with the general population (20.6 per 1,000 person-years versus 12.1 per 1,000 person-years). The 5-year accident risk in patients aged 18 to 69 years with syncope was 8.2%, compared with 5.1% in the general population.
The risk of syncope recurrences is highest in the first year after the initial event, then it gradually tapers off and reaches a plateau after 5 years. About one-quarter of syncopal episodes while driving remain undiagnosed. The annual recurrence rate of undiagnosed syncope (15–21%) lies in between the recurrence rate of neurally mediated syncope and syncope of other aetiologies, but the actual risk of recurrence of syncope while driving is low at <1.1% per year, which is similar to the risk of SCI in the RH formula.15 It follows that the highest risk of recurrence while driving actually resides with neurally-mediated syncope, but the actual RH of this type of syncope rarely reaches the threshold of unacceptable societal risk (0.005% per year). Bradyarrhythmic syncope recurrences are usually mitigated by implantation of a permanent pacemaker. AVNRT and AVRT are effectively treated and even cured with radiofrequency ablation. The case of VT/VF will be discussed below, under the headings for ICDs.
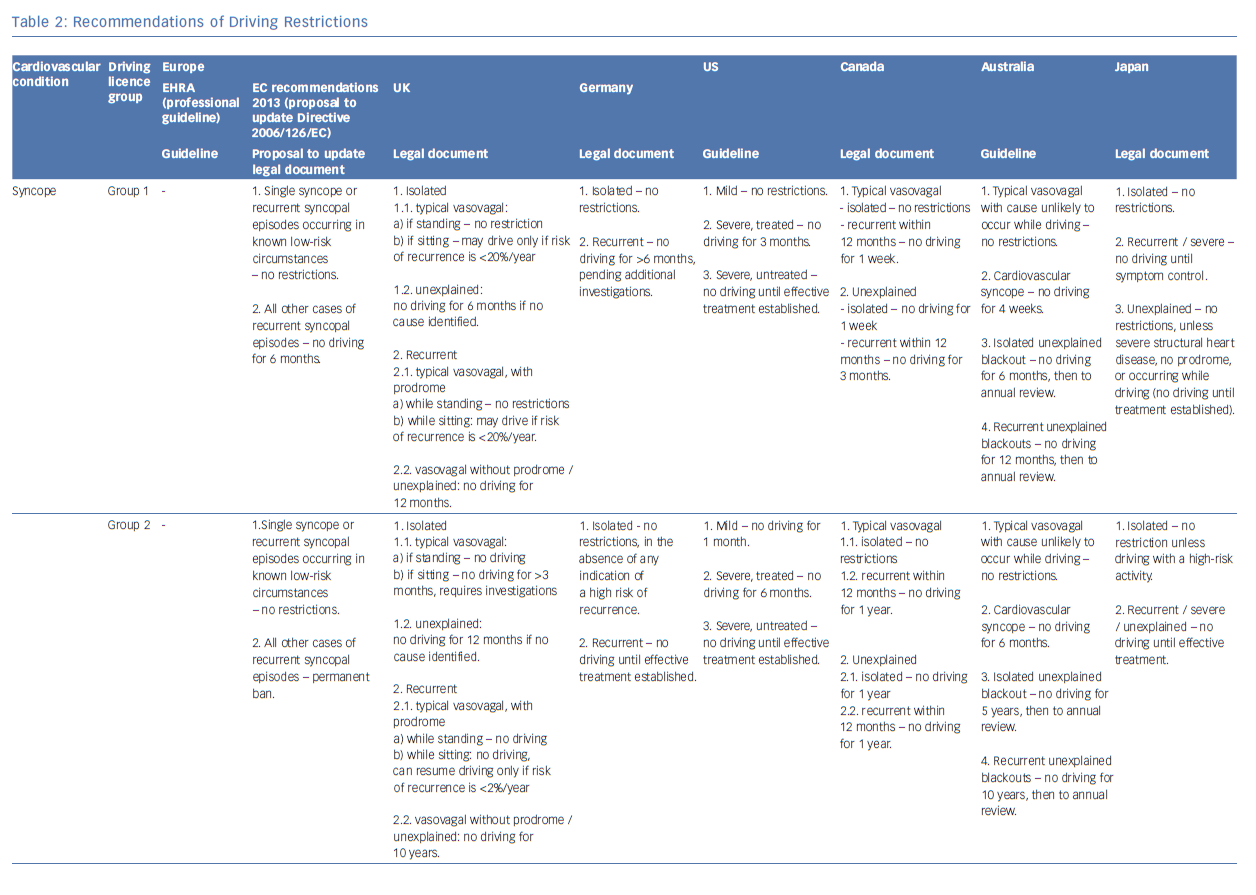
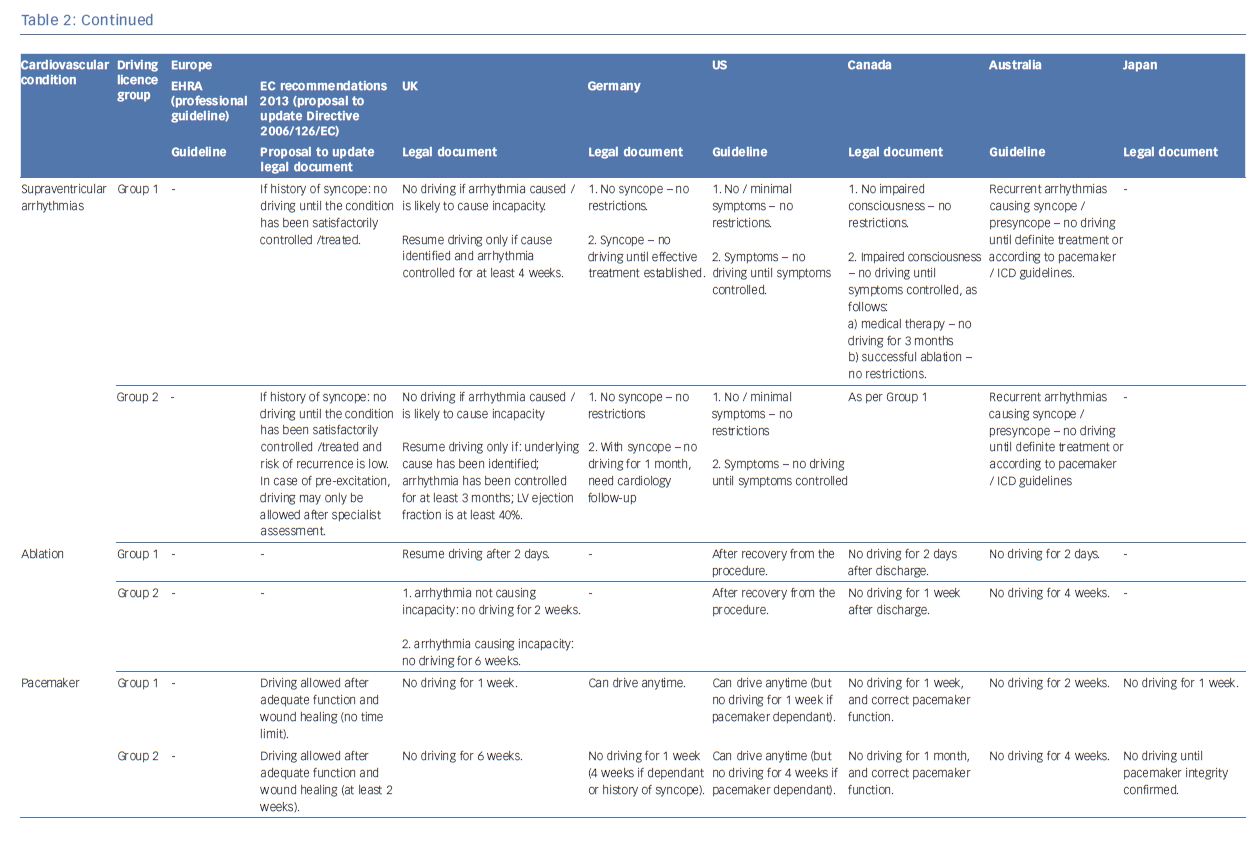
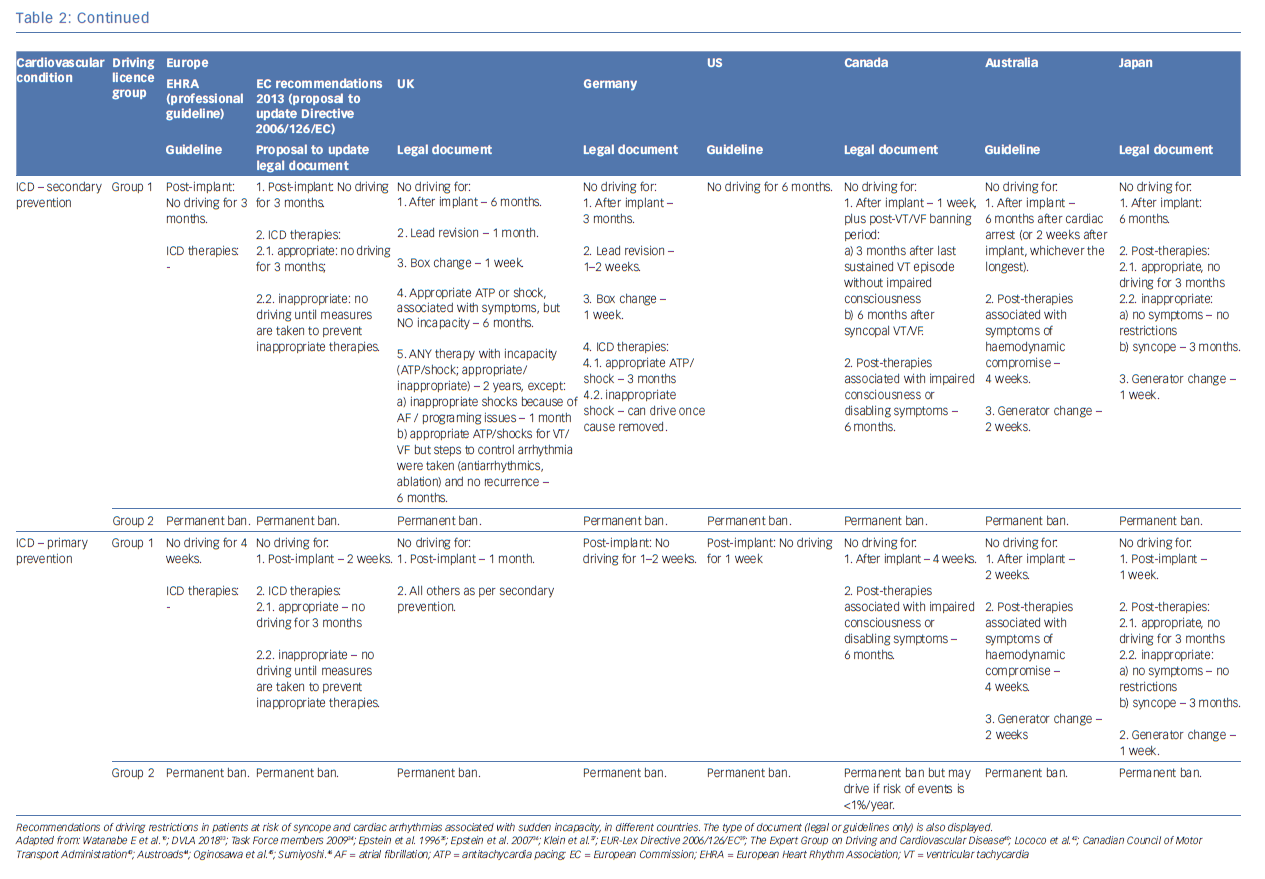
See Table 2 for full details of syncope guidance depending on country or guideline document. We endorse the recommendations valid in the UK on driving restrictions for patients with syncope, supraventricular tachycardia, following ablation procedures and with pacemaker devices. Our personal opinion regarding private drivers with ICDs will be discussed briefly below.
ICDs
In 2011, around 400,000 ICDs were implanted each year worldwide, two-thirds of which were new implants.19 ICDs are implanted for primary or secondary prevention of SCD. Primary prevention refers to patients who have never had but are at risk of having a VT/VF event. Secondary prevention refers to patients who have had a VT/VF event. There are a variety of conditions that may predispose a patient to SCD and each carries a particular risk. In the adult population, the majority of SCD events – approximately 80% – appear in patients with coronary artery disease.20,21
ICDs are effective in treating sudden ventricular tachyarrhythmic events that can cause SCD. However, ICDs do not prevent such events. With VF, loss of consciousness is usual as the ICD typically takes 10–15 seconds to deliver therapy (longer for subcutaneous ICDs). As such, establishing the risk of syncopal events caused by VT/VF in patients with ICDs is important to assess the RH.
The average annual risk of shock while driving in patients with ICDs is approximately 1.5%.22 Studies have documented that the risk of syncope associated with appropriate ICD shocks in patients who have had an ICD implanted for secondary prevention ranges from 2.0% to 16.0% (average 11.2%).10 For primary prevention, the risk of syncope associated with appropriate ICD shocks ranges from 0.6% to 4.3% (average 1.6%).10 The Triggers of Ventricular Arrhythmias (TOVA) study suggested that the absolute risk of ICD shock for VT/VF within 1 hour of driving is approximately one episode per 25,116 person-hours spent driving. Interestingly, the increased risk of shock was observed primarily in the 30-minute period after driving (RR 4.46; 95% CI [2.92–6.82]) rather than during the driving episode itself (RR 1.05; 95% CI [0.48–2.30]).23
Fewer data are available regarding the risk of sudden incapacitation associated with inappropriate ICD shocks. Data from a study performed in Japan suggest that only 0.7% of patients who experience inappropriate ICD therapies also have syncope, e.g. because of fast AF resulting in syncope but terminated by ICD shock, or VF induced by inappropriate ICD shock-on-T-wave as a result of T-wave oversensing.24 The calculated RH for inappropriate ICD therapies associated with syncope was <0.0008% for both primary and secondary prevention ICD indications, leading the authors to conclude that inappropriate ICD shocks should not result in a driving ban.24
Current data suggest that there is an increased risk of ICD shocks early after ICD implantation – for both primary and secondary prevention – and following appropriate or inappropriate ICD shocks, but the risk rapidly diminishes over the next 6 months. Thijssen et al. analysed data from 2,786 patients with primary and secondary prevention ICDs. Using the societal threshold for the RH of 0.005%, the 95% CI of the annual RH following ICD implantation was always below the threshold for both primary and secondary prevention, suggesting that no specific period of restriction after implantation is appropriate for private drivers. Following appropriate ICD shocks – and using a historical estimated risk of syncope associated with appropriate ICD shocks of 31% – the 95% CI of the annual RH fell below the threshold at 6 months for primary prevention ICDs, and at 3 months for secondary prevention ICDs. For commercial drivers, the RH was always above the threshold, supporting a permanent driving ban.25 However, newer data on contemporary ICD patient populations with modern ICD programing – and a more contemporary estimated risk of syncope associated with ICD shocks of 14% – suggest that the RH falls below 0.005% only 1 month after appropriate shocks.26 Thijssen et al. also estimated the RH after inappropriate shocks, but they assumed that the risk of syncope associated with ICD shocks is identical (31%) regardless of whether the shock was appropriate or not, which likely resulted in significantly overestimated RH (the 95% CI of the annual RH fell below 0.005% at 1 month and 3 months for appropriate and inappropriate shocks, respectively).25 As mentioned, newer data suggest that driving restrictions may not be necessary after inappropriate shock therapy.24
It is important to realise though that there are several important limitations regarding the RH assessment in patients with ICDs. First, as discussed, the RH threshold of 0.005% has been historically accepted for Canadian populations based on Canadian road traffic accident data from more than 30 years ago. Second, the risk of SCD and ICD shocks has been largely based on populations from the 1990s and early 2000s but there has been an almost 70% reduction of mortality in patients with coronary artery disease and heart failure in the last 20 years and a 44% reduction in SCD rates between 1995 and 2014 in patients with heart failure and reduced ejection fraction.27,28 These dramatic changes were a result of more effective drug treatment, e.g. angiotensin converting enzyme inhibitors, early revascularisation in patients with acute coronary syndromes, implementation of cardiac resynchronisation therapy, and so on. Indeed, in non-ischaemic dilated cardiomyopathy, the Danish Study to Assess the Efficacy of ICDs in Patients with Non-ischemic Systolic Heart Failure on Mortality (DANISH) failed to show a benefit of ICDs in reducing mortality, compared with the 11-year older Sudden Cardiac Death in Heart Failure Trial (SCD-HeFT). However, the overall absolute 5-year mortality rate in the DANISH was approximately 10% lower than in the SCD-HeFT.29,30 As such, the risk of sudden incapacity while driving in contemporary patients with ICD may well be lower than the historical data upon which the current recommendations and legislations are based would suggest.
Based on the summary above, we think that the current driving restrictions for patients with ICDs holding a group 1 driving licence are, in some cases, too restrictive. We propose that the following driving restrictions should suffice for these patients, if they drive in countries where the road safety statistics are similar to the countries mentioned in Table 2:
- after ICD implantation or box-change (both primary and secondary prevention) = 1 week;
- following appropriate ICD shock whether or not associated with incapacity = 1 month; and
- following inappropriate ICD shock whether or not associated with incapacity = no restriction if cause corrected.
Legislation and Disclosure of Patient Information
Driving Regulations and Expert Consensus Documents
In many countries driving regulations have evolved over time as new data on clinical outcomes have become available. For example, in the UK, driving with an ICD was initially completely prohibited. By 1994 driving was allowed 2 years after ICD implant and by 2000 the regulations evolved to allow driving 1 month after a primary prevention ICD and 6 months after a secondary prevention ICD.31–33 Regulations are made to provide a balance between the privilege of driving and the potential to harm others from driving. It can be argued that, based on cultural and social mentality, national legislation will find different levels of equilibrium between these two opposing forces. In addition to national regulations, professional bodies have published guidance relating to particular areas of interest, such as licensing in ICD patients.34–36 The different national regulations and physician recommendations are summarised in Table 2. In some areas there is general consensus on no professional driving for patients with ICDs, in other areas there is more variation.
In the UK, driving regulations are governed by the Driver and Vehicle Licensing Agency for England, Wales and Scotland, and Driver and Vehicle Agency for Northern Ireland.33 In Germany, assessment of fitness to drive is governed by the German Federal Highway Research Institute.37
In Europe in 1991, recognising substantial variation in the detail and implementation of driving regulation among EU Member States, the European Council established a Directive regarding the minimal physical and mental fitness standards for driving a vehicle, but the recommendations of this document for cardiovascular disease were vague. The Directive explicitly stated that driving is incompatible with the presence of “any disease capable of exposing [the driver] to a sudden failure of the cardiovascular system such that there is a sudden impairment of the cerebral functions”, and with the presence of “serious arrhythmias” (a condition left undefined), while patients with cardiac pacemakers may drive if adequate follow up and checks are established.38 The presence of ICDs or ablation of cardiac arrhythmias is not found anywhere in the Directive, which was issued before these interventions were widely implemented in clinical practice. The latest amendment of this Directive (Directive 2006/126/EC, Annex III), which is still in force, made no changes to these definitions.39 This Directive has been widely incorporated into several legal frameworks and was legally binding in some European countries. Fortunately, the European Council has undertaken efforts to update the Directive 2006/126/EC Annex III with more extensive, up-to-date and specific recommendations for driving in patients with cardiovascular diseases.40 These changes have already been implemented in those EU member countries where the prior legislation was in effect until very recently, e.g. Romania.41
The legal framework of driving restrictions in the US is highly variable between states as there is no over-ruling federal law governing licensing decisions on medically at-risk drivers. For example, some states have a Medical Advisory Board (MAB) to guide decisions, while others do not. In addition, in some states medical professionals review cases, while in others administrative staff perform reviews. In some states, MABs employ medical professionals (e.g. Maine, North Carolina), while in others it is administrative staff who employ medical professionals (e.g. Texas, Wisconsin). Other states have no MAB, but again it is either medical professionals who perform reviews (e.g. Oregon), or administrative staff (e.g. Ohio, Washington).42
In Canada, the individual provinces and territories can legally develop their own policies but for consistency a central body – The Canadian Council of Motor Transport Administrators – has established a Driver Fitness Overview Group to advise on uniform medical standards. These standards are highly detailed and, unusually, allow for the possibility of commercial driving in recipients of a primary prevention ICD, in subgroups where the annual risk of incapacitation is below 1%.43
In Australia, Austroads and the National Transport Commission have issued guidelines on driving in patients with cardiovascular diseases.44 In Japan, regulations for drivers with cardiovascular diseases are governed by a Road Traffic Act issued by the Japanese National Police Agency.10,45,46
Patient Confidentiality and Duty to Report Non-adherence
In general, it is a physician’s responsibility to be familiar with the regulatory framework in the country where they practise. They are responsible for informing the patient what regulations apply and whether the patient should be notifying the driving authorities of their condition.
Adherence with physician recommendations regarding driving is low in patients with ICD, with approximately one-third of patients not adhering to these recommendations.22 Patients frequently perceive the driving restrictions as a loss of independence and change in self-image. Often patients resume driving because of a misunderstanding about their condition and the risks involved, or because they think it is their decision not others to make.47 Education about the rationale for driving restrictions is important for ICD patients.
In a situation where a physician becomes aware that a patient is not adhering to the local driving code, an ethical issue arises about what to do. In the US, the recommended ethical action for doctors who are involved in the care of patients with conditions that constitute a ban from driving is to disclose that information to the police, after informing the patient, even if the patient refuses to obey.36 The reasoning is that ethical responsibilities of beneficence (do good and avoid evil) and non-maleficence (do no harm) take precedence over the principle of confidentiality in this setting. In Canada, disclosure of patients’ information by physicians is mandatory in most states, but not in all (for example, reporting is discretionary in Alberta, Nova Scotia and Quebec).11 In the UK, doctors should inform patients about conditions and treatments that might affect their ability to drive and remind them of their duty to tell the appropriate agency.33 If a patient refuses or is found not to have told the appropriate agency, doctors should ask for a patient’s consent to disclose information to the authorities, unless the information “is required by law or if it is not safe, appropriate or practicable to do so”.48 In Germany, because of confidentiality law, the doctor should only inform the patient regarding the loss of fitness to drive; informing the authorities is not permitted.37 In Japan, the doctor should advise patients not to drive if they have had syncope or are at risk of syncope. Also, the doctor is recommended to advise about conditions or treatments that might affect the patient’s ability to drive to the National Public Safety Commission (Watanabe E, personal communication).
Conclusion
Driving regulations for patients at risk of syncope and cardiac arrhythmias associated with sudden incapacity attempt to balance the perceived RH against protection of individual freedom and the right to drive. There is significant national variation in regulation and the approach to its implementation.
Much of the scientific data that back up current recommendations are historical and may not accurately reflect changes in vehicles and the driving environment, along with possible changes in societal acceptance of risk. In future, for private drivers, a method to estimate the individual RH while driving – based on individual assessment of the time spent behind the wheel, age, driving profile, car safety, and so on – may prove useful. The development of new technologies such as driverless vehicles may have an impact on society’s willingness to accept excess risk as a result of medical conditions.