Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia and is characterised by rapid and irregular heart rates. It is a lifethreatening condition present in up to 1.5 % of the population and accounts for approximately 15 % of all stroke events.1 While relatively unusual in those under 55 years, its incidence increases substantially with age, particularly between the ages of 65 and 80 years,2 and its incidence and prevalence are rising at rates that are not completely explained by an aging population.3 Patients with AF have an approximately five-fold increased risk of stroke compared with those without AF.4 Consequently, AF and AF-related stroke are a major burden on healthcare systems in Europe and the US.5,6 Stroke risk can vary up to 20-fold between patients with AF, depending upon the presence or absence of clinical risk factors. These risk factors were collated in the CHADS2 scoring scheme for stroke risk and subsequently revised in the CHA2DS2-VASc scheme.7 In recent years a large number of interesting studies have investigated the use of new oral anticoagulants for stroke prevention in patients with AF. This article aims to outline the usage, efficacy and safety of nonvitamin K antagonist oral anticoagulants (NOACs), to describe the recent Effective Anticoagulation with Factor Xa Next Generation in Atrial Fibrillation – Thrombolysis In Myocardial Infarction Study 48 (ENGAGE AF-TIMI 48) and its implications for stroke prevention in AF, and to consider future directions in oral anticoagulation in AF, based on a satellite symposium held at the Cardiostim/EHRA Europace Congress in Nice in June 2014.
The Evolving Treatment Landscape for Atrial Fibrillation in Europe – What Choice for Stroke Prevention?
For a number of years, vitamin K antagonist (VKA) therapy has been used for stroke prevention in patients with AF. In terms of stroke prevention, it is superior to aspirin and its benefit is not offset by the occurrence of major bleeding.8 In the 1990s, the use of warfarin increased substantially, following a number of clinical trials that demonstrated a significant reduction in stroke risk compared with placebo.9 However, VKA therapy has several limitations that make it difficult to use in clinical practice. These include an unpredictable pharmacokinetic response, narrow therapeutic index, slow onset and offset of action, the need for routine coagulation monitoring and frequent dose adjustment, and numerous food–drug and drug–drug interactions.10–12
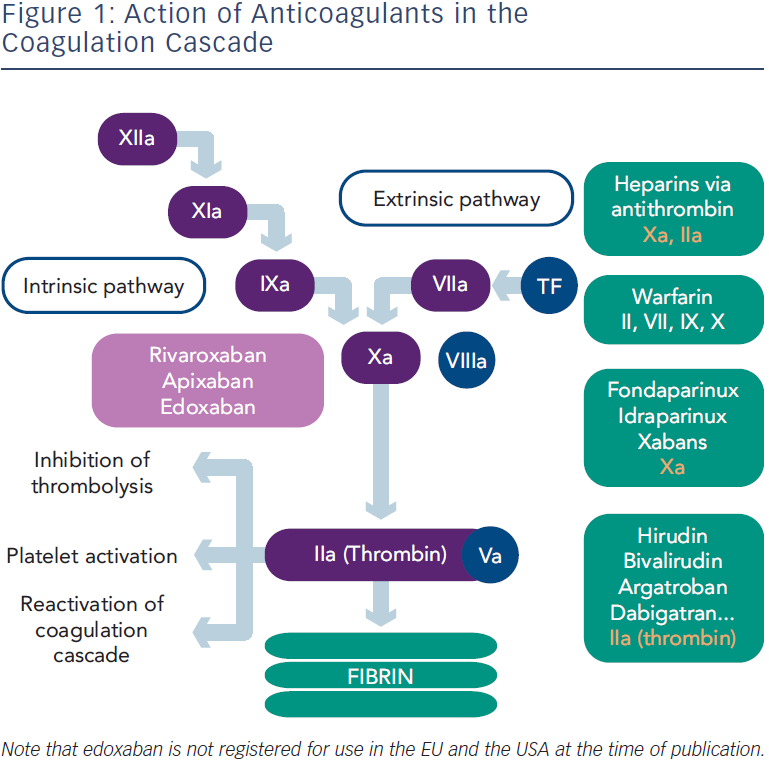
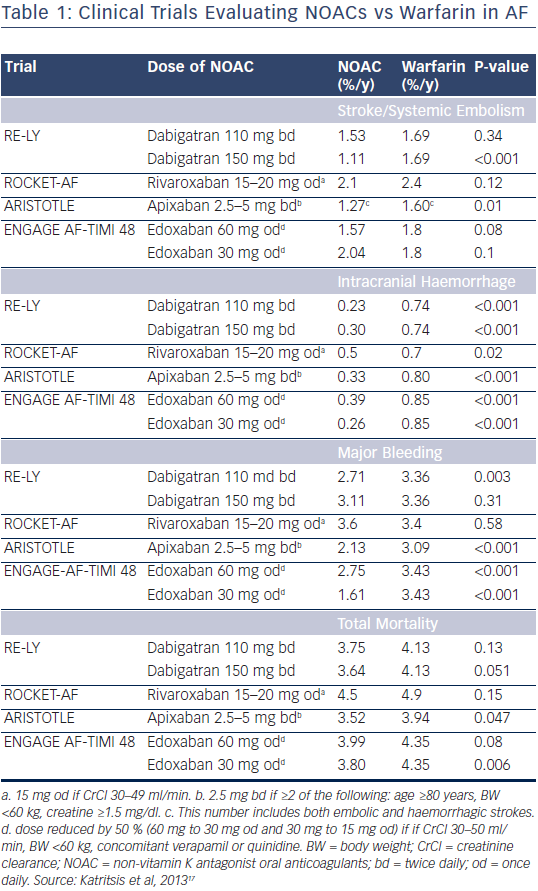
In recent years, NOACs that directly inhibit thrombin or factor Xa have been developed for stroke prevention in patients with AF (see Figure 1). Investigations have been performed with one direct thrombin inhibitor (dabigatran) and three factor Xa inhibitors (rivaroxaban, apixaban and edoxaban). The first study to be completed was the Randomised Evaluation of Long Term Anticoagulant Therapy With Dabigatran Etexilate (RE-LY) trial in 2009, which showed that dabigatran was non-inferior to warfarin for prevention of stroke and systemic embolism, and also associated with lower rates of major bleeding.13 Following this pivotal clinical trial, three factor Xa inhibitors have demonstrated non-inferiority to warfarin in terms of prevention of stroke and systemic embolism: rivaroxaban in the Rivaroxaban-Once Daily, Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF) trial,14 apixaban in the Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation (ARISTOTLE) trial15 and edoxaban in the ENGAGE AF-TIMI 48 trial.16 A summary of the major findings of clinical trials to date is given in Table 1.17 At present, three NOACs (dabigatran, rivaroxaban and apixaban) have been approved for stroke prevention in patients with AF in Europe.
A recent meta-analysis of the clinical trials with NOACs in patients with AF found they had a favourable risk-benefit profile overall compared with warfarin, with significant reductions in stroke and systemic embolism, intracranial haemorrhage, mortality, and a reduction in the risk of major bleeding, although there was as an increased risk of gastrointestinal bleeding.18 Although all four NOACs significantly reduced the risk of haemorrhagic stroke, reduction in ischaemic stroke was only seen with dabigatran 150 mg twice daily.13 There was also a degree of variation between the NOACs vs warfarin in gastrointestinal bleeding, with an increase with dabigatran 110 mg and 150 mg twice daily, rivaroxaban and high-dose edoxaban 60 mg once daily treatment, whereas no increase with apixaban and lowdose edoxaban 30 mg once daily. In addition, dabigatran 110 mg and 150 mg twice daily and low-dose edoxaban 30 mg once daily were associated with a decrease in all-cause mortality.13,16 As the majority of patients with AF are aged over 75 years, a recent meta-analysis analysed data from participants in clinical trials aged 75 years and over, and concluded that NOACs were associated with equal or greater efficacy than conventional therapy and did not cause excess bleeding in this population.19
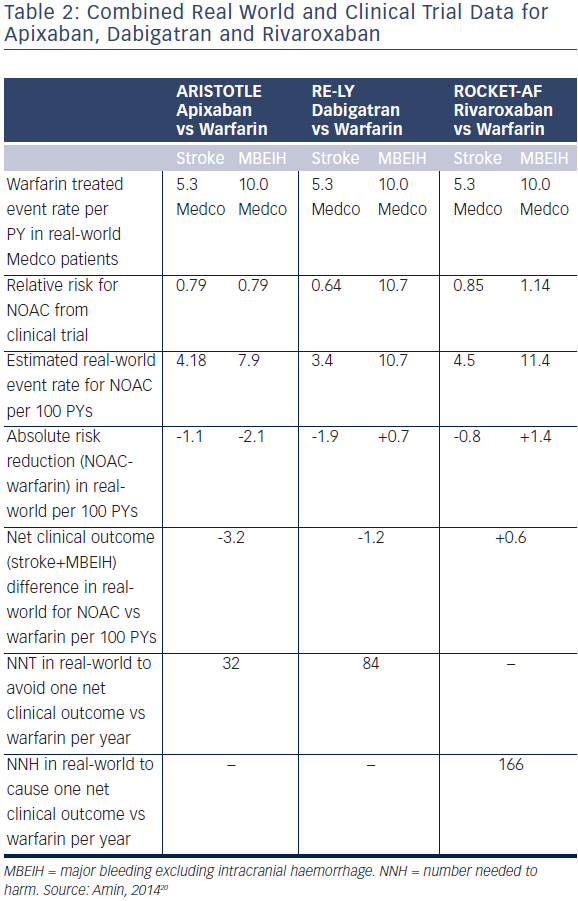
However, when comparing efficacy and safety observations with the different NOACs it is important to consider that no head-to-head comparison of the NOACs has been performed, and also that the four pivotal trials had different designs and patients with varying risk of stroke, making comparisons between the studies highly confounded. Edoxaban has not yet received approval for use in Europe, however dabigatran, rivaroxaban and apixaban are in clinical use in many countries. A recent US study combined clinical trial data and a real world sample, taken from Medco patients during 2007–2010 (see Table 2).20 The study concluded that if relative risk reductions from randomised clinical trials persist in the real world, apixaban gives the greatest clinical benefit vs warfarin of the three NOACs in terms of stroke and major bleeding excluding intracranial haemorrhage events avoided.
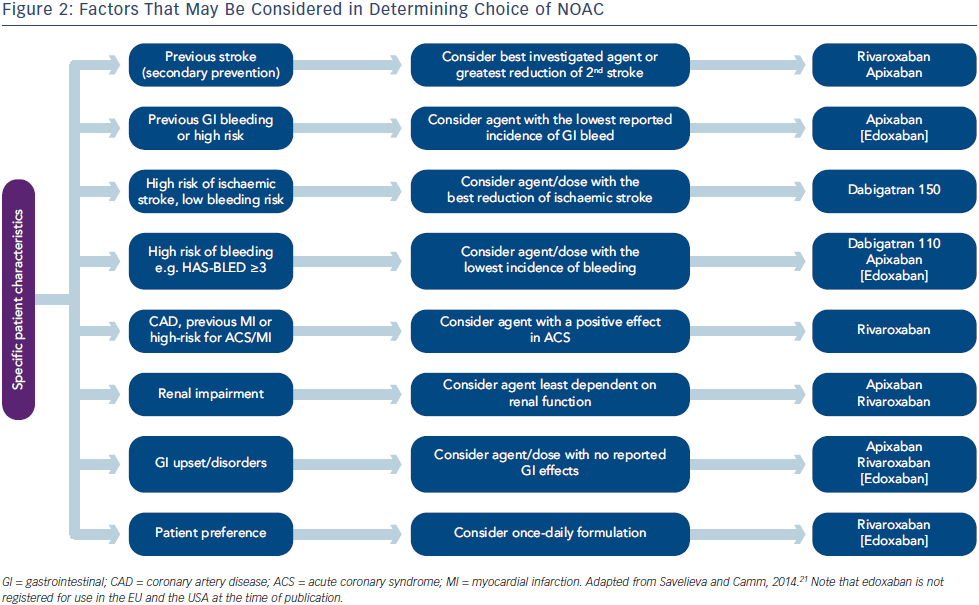
Alongside these clinical trial data, an important issue in choosing between the NOACs is the potential for patient reluctance in switching from a once-daily VKA to a twice-daily NOAC, rather than a once-daily NOAC. Treatment discontinuation is also of relevance and apixaban and edoxaban treatment were both associated with a lower rate of discontinuation compared with warfarin.15,16 Therefore, considering these aspects of NOAC treatment, it has been suggested that the choice of NOAC be guided by specific patient characteristics (see Figure 2).21
An additional treatment option for patients with non-valvular AF is percutaneous closure of the left atrial appendage (LAA). A multicentre, non-inferiority trial randomised 707 patients to percutaneous closure of the LAA and subsequent discontinuation of warfarin or warfarin treatment. Efficacy was assessed by a primary composite endpoint of stroke, cardiovascular death and systemic embolism. The efficacy of percutaneous closure of the LAA with this device was noninferior to that of warfarin therapy. Although there was a higher rate of adverse safety events in the intervention group than in the control group, events in the intervention group were mainly a result of peri-procedural complications.22 Longer term (mean 2.3 years) data from this study confirmed that LAA closure was noninferior to systemic anticoagulation with warfarin. There were more primary safety events in the LAA closure group (5.5 % per year; 95 % confidence interval [CI], 4.2 %–7.1 % per year) than in the control group (3.6 % per year; 95 % CI, 2.2 %–5.3 % per year; relative risk, 1.53; 95 % CI, 0.95–2.70).23 This is therefore a recommended treatment option for patients at high risk of stroke and in whom longterm treatment with oral anticoagulation is contraindicated.
In conclusion, the landscape of atrial fibrillation and antithrombotic treatment has dramatically evolved in recent years, as a result of the availability of NOACs. As no head-to-head comparison of NOACs has been performed, it has been suggested, when choosing a NOAC therapy, to take into account individual patient factors and medical history.
ENGAGE AF-TIMI 48 – Implications for Stroke Prevention in Atrial Fibrillation
An extensive phase II dose-ranging program was undertaken with edoxaban to inform the design of phase III studies. A phase II study in 1,146 patients with non-valvular AF compared a number of edoxaban dosing strategies (30 mg once daily, 60 mg once daily, 30 mg twice daily and 60 mg twice daily) with open-label, dose-adjusted warfarin. Edoxaban 30 mg and 60 mg once daily both had a similar safety profile to warfarin and were both associated with less bleeding than edoxaban 30 mg twice daily.24 Therefore in the phase III ENGAGE AF-TIMI 48 study, only once-daily edoxaban treatment was compared with warfarin.
ENGAGE AF-TIMI 48 was a double-blind, double-dummy, randomised, international study in patients with confirmed non-valvular AF.16 The study compared two once-daily edoxaban regimens with doseadjusted warfarin (international normalised ratio [INR] 2.0–3.0).25 The edoxaban dose was halved in patients at risk of edoxaban overexposure or bleeding at randomisation or at any time during the study (dose reduction criteria: body weight ≤60 kg, creatinine clearance 30–50 ml/min, or concomitant treatment with a strong P-glycoprotein inhibitor). In addition to dose reduction, comprehensive assessment of patient INR values and a transition period at study end were undertaken.25 Patients (n=21,105) who had AF documented by electrocardiogram in the previous 12 months and a CHADS2 score ≥2, were randomised 1:1:1 to low-dose edoxaban 30 mg once daily (or reduced to 15 mg once daily), high-dose edoxaban 60 mg once daily (or reduced to 30 mg once daily) or dose-adjusted warfarin (INR 2.0- 3.0). Randomisation was stratified by CHADS2 score and anticipated drug exposure. The primary efficacy endpoint was a composite of stroke (ischaemic or haemorrhagic) and systemic embolic events, and the non-inferiority boundary for the upper limit of the 97.5 % CI was 1.38. The primary safety endpoint was major bleeding.
Of patients enrolled, 99.6 % received the drug. Completeness of follow-up was 99.5 %, and 99.1 % of those enrolled were present at final visit. Only 8.8 % of patients per year came off study drug, 0.9 % withdrew consent and only one patient was lost to follow-up. Compared with other NOAC trials in AF, ENGAGE AF-TIMI 48 was the longest trial with a median follow up of 2.8 years, and had the highest median time in therapeutic range (TTR) of 68.4 %.16
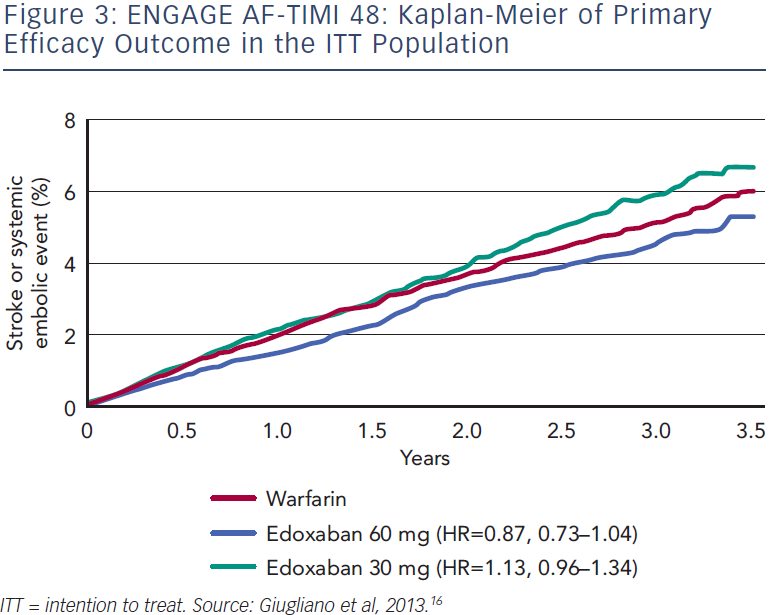
The primary efficacy analysis in the modified intention to treat (mITT) on-treatment population found stroke or systemic embolism at a rate of 1.50 % per year in the well-controlled warfarin group, 1.18 % per year in the high-dose edoxaban 60 mg once-daily group (hazard ratio [HR] 0.79; p<0.001 for non-inferiority; p=0.02 for superiority) and 1.61% per year in the low-dose edoxaban 30 mg once-daily group (HR 1.07; p=0.005 for non-inferiority; p-value not significant for superiority). The Kaplan-Meier curve for the primary efficacy outcome in the intention to treat (ITT) population showed a lower rate of stroke or systemic embolism in the high-dose edoxaban group from early in the study (see Figure 3). Key secondary outcomes included a lower rate of haemorrhagic stroke with both doses of edoxaban (HR 0.33 and 0.54 for low-dose and high-dose edoxaban, respectively, vs warfarin). In terms of ischaemic stroke, no difference was seen between highdose edoxaban and warfarin, but a statistically significant increase was seen with the low dose (HR 1.41; p<0.001). All-cause mortality was significantly reduced with low-dose edoxaban vs warfarin (HR 0.87, p=0.006) and a non-significant reduction was seen with the high-dose edoxaban group. However, cardiovascular mortality was significantly reduced with both low-dose and high-dose edoxaban regimens vs warfarin (HR 0.85; p=0.008 and HR 0.86; p=0.013,respectively). There was also no statistically significant difference in the rate of myocardial infarction between the low-dose and highdose edoxaban groups and warfarin.16
In terms of safety outcomes, compared with warfarin, the annualised rate of major bleeding was significantly lower with both high-dose edoxaban (2.75 % vs. 3.43 %; HR 0.80; p<0.001) and also low-dose edoxaban (1.61 % vs. 3.43 %; HR 0.47; p<0.001). Significant reductions in fatal bleeding and intracranial haemorrhage were also observed with both edoxaban regimens vs warfarin (p≤0.006 for all comparisons). A significantly lower rate of gastrointestinal bleeding was seen in the low-dose edoxaban group vs warfarin (HR 0.67; p<0.001), but a significantly higher rate occurred in the high-dose edoxaban group (HR 1.23; p=0.03). Analysis of three net clinical outcomes (stroke/ disabling stroke, systemic embolism, major/life-threatening bleeding and all-cause mortality) showed that all were significantly reduced with both dosage regimens of edoxaban compared with warfarin (p<0.01 for all comparisons).16
At the end of the study, patients were extensively monitored during transition to open-label oral anticoagulant therapy. Patients were either transitioned to open-label VKA, overlapping active low-dose edoxaban and VKA for 2 weeks or until the INR reached 2.2, whichever came first; or transitioned to an open-label NOAC. In the case of an INR <2.0, NOAC treatment was initiated. If the INR was ≥2.0, INR measurements were repeated until the INR dropped below 2.0, then the NOAC was initiated. This approach resulted in a similar incidence of stroke or systemic embolism in all three treatment groups (0.16 %, 0.15 % and 0.15 % with warfarin, high-dose and low-dose edoxaban, respectively), and also with the incidence of major bleeding through 14 days (0.13 %, 0.09 % and 0.11 % with warfarin, high-dose and lowdose edoxaban, respectively).16
In a recent subanalysis of the ENGAGE AF-TIMI 48 study, the efficacy of edoxaban was assessed relative to warfarin according to AF subtype.26 The guidelines state that no difference in stroke risk exists between different AF subtypes. The likelihood of stroke and systemic embolus was lower in paroxysmal AF subtypes compared with other forms of AF. In addition, the efficacy and safety of edoxaban compared to warfarin was preserved across the subtypes of AF. In terms of bleeding, no differences were seen according to AF subtype.26
In summary, compared with well-managed warfarin, both once-daily highdose and low-dose edoxaban were non-inferior for prevention of stroke and systemic embolism, with a trend to decreased stroke and systemic embolism in the high-dose edoxaban ITT analysis. Both edoxaban regimens also reduced major bleeding, intracranial haemorrhage, haemorrhagic stroke and cardiovascular death compared with warfarin. There was also no excess in stroke or bleeding during the transition to oral anticoagulant at the end of the trial. A recent review suggested that, given the limitations with long-term warfarin therapy, once-daily edoxaban may provide a convenient long-term alternative for patients.27
Future Directions for Oral Anticoagulants in Atrial Fibrillation – Where Do We Go From Here?
The availability of NOACs has enabled clinicians to tailor therapy to the patient, and it has been suggested that the availability of four NOACs has left the physician spoilt for choice.28 In order to facilitate future use of NOACs, there is a need for a consensus on the terminology used to describe them. The acronym NOAC is no longer appropriate, since these agents are now neither new nor novel, and new terminology is increasingly used in different countries. However, an acronym change may result in confusion when encountered in older publications or guidelines. It has therefore been recommended that the NOAC acronym should refer to non-VKA oral anticoagulants.29
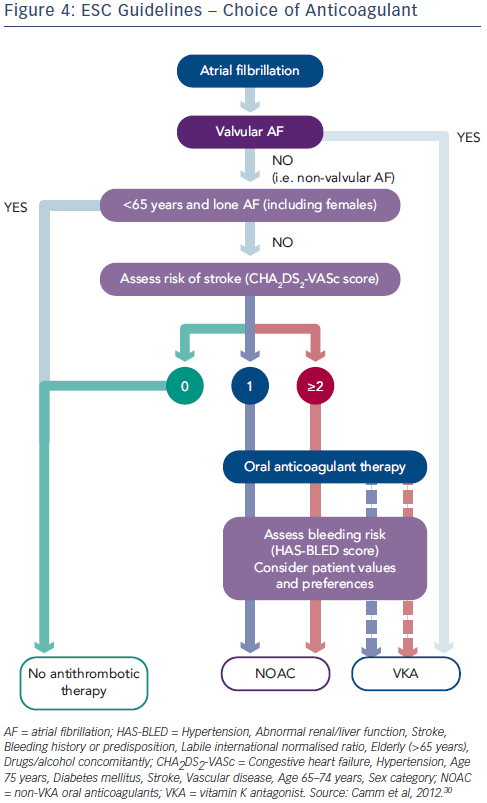
In the past, European guidelines focused on identifying ‘high risk patients’ as VKA therapy was considered troublesome and inconvenient. However, the ESC guidelines have evolved to reflect the efficacy and safety of the NOACs and a recent 2012 update recommended a practice shift towards greater focus on identification of ‘truly low risk’ patients with AF (aged <65 years and CHADS2 score of 0 for men or 1 for women), for whom antithrombotic therapy is not needed (see Figure 4).30 Effective stroke prevention can then be given to those patients at higher risk.
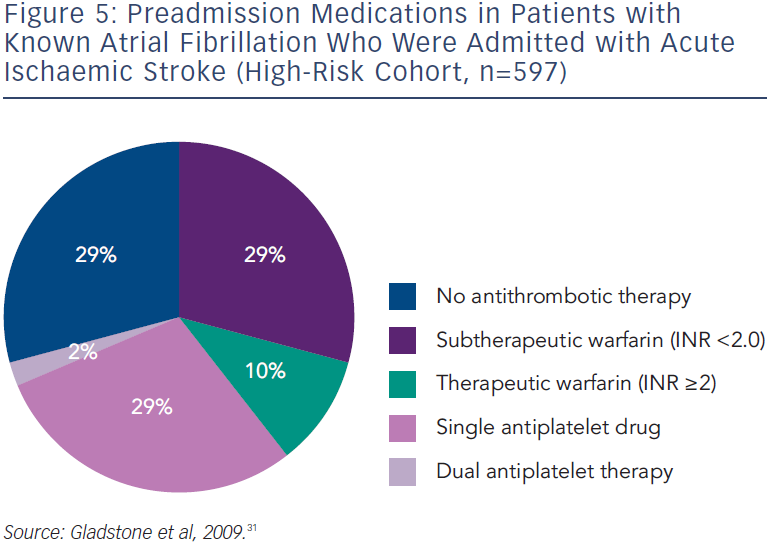
Given the advances in AF management and the updated ESC guidelines, there is a need for the systematic collection of information on the current management and treatment of AF. Data from a Canadian 2009 prospective stroke registry of 597 patients with AF at high risk of stroke showed that the majority of stroke events occurred in patients who were not within the therapeutic INR range, and only 10 % of patients with acute stroke and known AF had INR values ≥2.0 at admission (see Figure 5).31 Aspirin, which is now known to be of limited efficacy in preventing stroke in AF,32 is still in widespread use.33 There is a need to identify and treat the untreated and switch sub-optimally managed patients to NOACs. The use of NOACs requires new approaches in many aspects of daily use and in specific clinical situations, including renal impairment and urgent surgical interventions. The European Heart Rhythm Association (EHRA) has also published a comprehensive guide detailing practical aspects of use of NOACs.34
In Europe, large registry studies investigating the use of NOACs in real-world settings are ongoing: the Global Registry on Long-Term Oral Antithrombotic Treatment in Patients with Atrial Fibrillation (GLORIAAF),35 the Global Anticoagulant Registry in the Field (GARFIELD),36 the Eurobservational Research Programme Atrial Fibrillation (EORP)37 and Prevention of Thromboembolic Events – European Registry in Atrial Fibrillation (PREFER in AF).38 Preliminary data from the GARFIELD study indicate that the proportion of patients receiving anticoagulant therapy varies widely between countries, from less than 30 % in China to more than 90 % in Italy. The registry also found that only 57 % of patients take anticoagulant therapy, and that a minority (43 %) achieved adequate INR control over the first 12 months.39 Recently, data have been published from the smaller PREFER in AF registry that examined differences among western European countries.38 The proportion of patients taking VKAs varied between 86.0 % (France) and 71.4 % (Italy). Warfarin was used predominantly in the UK and Italy (74.9 % and 62.0 %, respectively), phenprocoumon in Germany (74.1 %), acenocoumarol in Spain (67.3 %), and fluindione in France (61.8 %). The major sites for INR measurements were biology laboratories in France, anticoagulation clinics in Italy, Spain and the UK, and the physicians’ offices or self-measurement in Germany.40
The EORP-AF registry found that oral anticoagulants were used in 80 % of patients, an increase over the last decade, but were predominantly VKAs (71.6 %), with NOACs being used in only 8.4 %. Other antithrombotics (mostly antiplatelet therapy, especially aspirin) were still used in one-third of patients, and no antithrombotic treatment in 4.8 %. Oral anticoagulants were used in 56.4 % of patients with CHA2DS2-VASc score of 0, with 26.3 % having no antithrombotic therapy. The study concluded that compliance with the treatment guidelines for patients with the lowest and higher stroke risk scores remains suboptimal.37 At present, there are no laboratory tests to assess the adherence of patients treated with NOACs.
The global position is more worrying. The RE-LY AF registry enrolled 15,400 patients at presentation to an emergency department at 164 sites and found a large global variation in AF management. The use of oral anticoagulation among patients with a CHADS2 score of ≥2 was greatest in North America (65.7 %) but was only 11.2 % in China. If VKAs are used, good quality anticoagulation control is essential. It is widely recognised that TTR is the best measure of assessing anticoagulant quality control. The mean TTR was 62.4 % in Western Europe and 50.9 % in North America, but only between 32 % and 40 % in India, China, Southeast Asia, and Africa.41 It is evident, therefore, that wider education regarding stroke prevention in AF is needed.
The individual patient’s values and preferences regarding stroke prevention and bleeding in AF are also important in the decision of which therapy to use. A recent study found considerable inter-patient variability in opinions. Overall, patients required at least a 0.8 % annual absolute risk reduction and 15 % relative risk reduction in the risk of stroke in order to agree to initiate antithrombotic therapy, and patients were willing to endure 4.4 major bleeds in order to prevent one stroke. In addition, 12 % of patients were medication averse and were not willing to consider antithrombotic therapy; even if it was 100 % effective in preventing strokes.42
A growing body of data is becoming available regarding the long-term efficacy and safety of NOACs. A recent US Food and Drug Administration (FDA) study in Medicare patients comparing dabigatran with warfarin, found there was no risk of increased MI with dabigatran. The latter was associated with a lower risk of clot-related strokes, bleeding in the brain, and death, but an increased risk of major gastrointestinal bleeding compared with warfarin.43 An ongoing clinical trial (Edoxaban vs Warfarin in Subjects Undergoing Cardioversion of Atrial Fibrillation [ENSURE-AF]) is currently comparing edoxaban to warfarin in subjects with AF who are undergoing cardioversion.44
Concluding Remarks
Stroke prevention is central in the management of AF. This is irrespective of rate or rhythm control and it is recommended that management is patient centred and symptom directed. The latest clinical trial of the NOAC edoxaban (ENGAGE AF-TIMI 48) demonstrated its non-inferior efficacy and improved safety vs well-managed warfarin. In addition to these clinical data with edoxaban and with the other NOACs, an increasing body of registry data is emerging that indicates VKA therapy is under-utilised and suboptimal in clinical practice, suggesting a switch to a NOAC may be of benefit for patients. There is therefore a need for increased education regarding the importance of anticoagulant use in AF.