A wide complex tachycardia (WCT) is simple enough to define: a cardiac rhythm with a rate >100 beats per minute and a QRS width >120 milliseconds (ms). Unfortunately, beyond this simple definition lies a complex differential diagnosis with prognoses ranging from utterly benign to potentially lethal, requiring treatment strategies ranging from medications to emergent non-sedated cardioversion and implantable cardioverter defibrillator (ICD) implantation (see Table 1). Practically, however, the differential diagnosis typically devolves to the question of ventricular tachycardia (VT) versus supraventricular tachycardia (SVT) with aberration. An intelligent, organised approach to WCTs is crucial to all practitioners responsible for the interpretation of an electrocardiogram (ECG), whether in emergency medicine, cardiology or primary care.
Aberrancy – From A to V
Via Normal Conduction System with Aberrancy
There exist only four ‘aberrant’ ways an atrial impulse can conduct over the atrioventricular (AV) node (AVN) and His-Purkinje system (HPS) to the ventricles: right bundle branch block (RBBB), RBBB with either left or right hemiblock and left bundle branch block (LBBB). As a first approximation, if the QRS complex during WCT cannot be resolved as using one of these four routes, the ventricle must be activating outside the specialised conduction tissue, limiting the differential to VT or SVT with ventricular activation over an AV accessory pathway (AP). Rarely, do patients with unusual hypertrophy patterns or repaired congenital heart disease have bizarre, wide QRS patterns during sinus rhythm; SVT in these patients will thus be similarly bizarre, potentially causing an SVT to appear most unusual for what is otherwise ‘normal’ conduction. Though this group of patients is growing in size, it remains a small proportion of all WCTs.
Via Pathway
Extranodal APs are abnormal muscle-to-muscle connections between atrium and ventricle across what should be the complete electrical insulation of the mitral or tricuspid valve annulus. As a result of their locations, ventricular activation during sinus rhythm occurs basally and epicardially. Since the AVN/HPS are also activated with each atrial impulse, the resulting QRS is usually a fusion of activation, with the initial component arising from the AP (the delta wave) and the latter part of the QRS a fusion between normal activation and the continuing wavefront from the AP. During atrial fibrillation, this manifests as irregular wide complex beats with variable QRS width caused by varying proportions of conduction over the AVN/HPS and pathway. Rare APs also exist between the specialised conduction tissues and the ventricle, but are infrequent enough to place them outside the scope of this review.
Via Pacemaker
In some ways, a pacemaker can be thought of as an accessory pathway inserting at the site of the ventricular lead(s). Depending on the lead’s tip location, a host of activation sequences are possible, especially if the HPS is also contributing to activation. Obviously, the presence of a pacing device on physical examination is a strong clue to this possible diagnosis. Modern bipolar pacemakers use so little voltage, confined to such a small area, that pacemaker spikes are frequently difficult to discern or are even invisible on the ECG. Modern ECG systems compensate for this by adding artificial pacing spikes when they detect the frequency characteristics of a pacing output, but this feature is imperfect in its ability to detect these impulses.
Do We Need an Algorithm at All?
Pre-test Probability
Multiple prior series have shown that, due to prevalence alone, the pre-test probability of a WCT being VT is in excess of 80 %.1–4 That is to say that if a reader simply declares all WCTs to be VT, that irresponsible individual will still be correct four out of five times. Furthermore, if the patient is known to have prior myocardial infarction, and symptoms of tachycardia did not begin until some time after the infarction, the odds of a WCT being VT exceed 90 %.3,4 The bar is thus set high for a differentiating algorithm to significantly improve on this accuracy in revealing the true diagnosis of WCT.
Importance of the Diagnosis
At Presentation
The primary goal of a correct diagnosis at presentation is the avoidance of harm. An SVT incorrectly believed to be VT may be treated with amiodarone or electrical cardioversion – not optimal therapy, but not harmful. If the presenting rhythm was instead atrial flutter, cardioversion in an unanticoagulated patient will incur a 1.5 % risk of stroke, harming one in every 66 patients.5 Worse still is the patient with VT treated as SVT. In this case, agents with negative inotropic effects such as verapamil or diltiazem may be used to control the presumed SVT. In one series, 100 % of patients given verapamil for an inappropriate diagnosis of SVT had haemodynamic deterioration.1 This mistake must thus be avoided if at all possible.
In Later Management
The more insidious consequences of misdiagnosed arrhythmia are found in the long term management of such patients. Incorrectly diagnosing an SVT as VT may result in the patient receiving long-term amiodarone therapy, or even an implantable defibrillator with repeated generator changes ahead. Regardless of which rhythm is misdiagnosed, potentially curative catheter-based therapies may be inappropriately withheld from the patient.
Common Mistakes in Diagnosis
It is a common misconception that a haemodynamically stable patient with minimal symptoms during a WCT episode must have SVT.2 The converse is likewise untrue. Often adding to this assumption is a belief that termination by adenosine or verapamil proves SVT. Again, this assumption can lead to misdiagnosis as some VTs are responsive to one or both of these agents. Fortunately, the vast majority of these VTs are not associated with significant structural heart disease and sudden death with episodes is extraordinarily rare.
Maintaining Focus on the Patient
The exercise of trying to apply algorithms to diagnose a WCT can distract a providers’ attention from the patient. Immediate cardioversion of the haemodynamically unstable patient with WCT should be the first thought, and only when some measure of stability has been achieved should a provider delay care to look up and apply the various algorithms described below.1
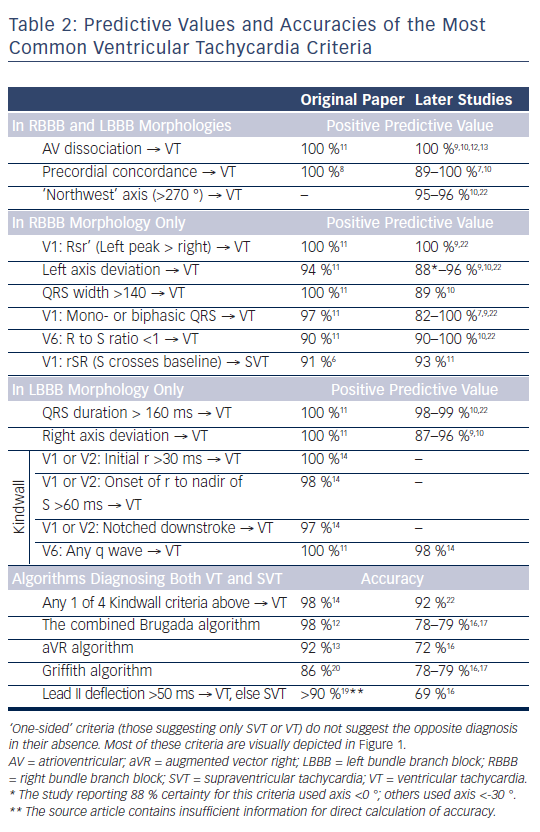
Principles of Ventricular Tachycardia Discrimination
Basis and Goals of Ventricular Tachycardia Criteria
In contrast to the SVTs described above, with relatively constrained modes of ventricular activation, VT can originate from literally any location within the ventricular myocardium or its specialised conduction tissues. As a result of this, VT can appear identical to any of the wide complex SVTs above and it is nearly impossible to say with absolute certainty, from an ECG of free-running tachycardia alone, that a wide complex rhythm must be supraventricular. As such, most algorithms seeking to discriminate the two entities focus on identifying characteristics unique to VTs – that is to say, those with high specificity for VT – and presume all else to be SVT until proven otherwise; the Griffith algorithm is the notable exception to this approach. Algorithms have been unable to reliably distinguish between VT and pre-excited SVT, since initial ventricular activation in the latter is (like most VTs) extrinsic to the normal conduction system. Since pre-excited SVTs comprise such a small proportion of WCTs, they will not be considered further in this discussion.
Defining Morphology
One of the first determinations in looking at a WCT should be its bundle branch block morphology. Ordinarily, the term bundle branch block (BBB) implies failure to conduct over one or more of the specialised fascicles, but in discussion of WCT, the question is designed more to evaluate which chamber activates last. This is done by looking at the terminal deflection of lead V1. A LBBB WCT is one with a terminal negative deflection in V1 and RBBB is then a positive terminal deflection.
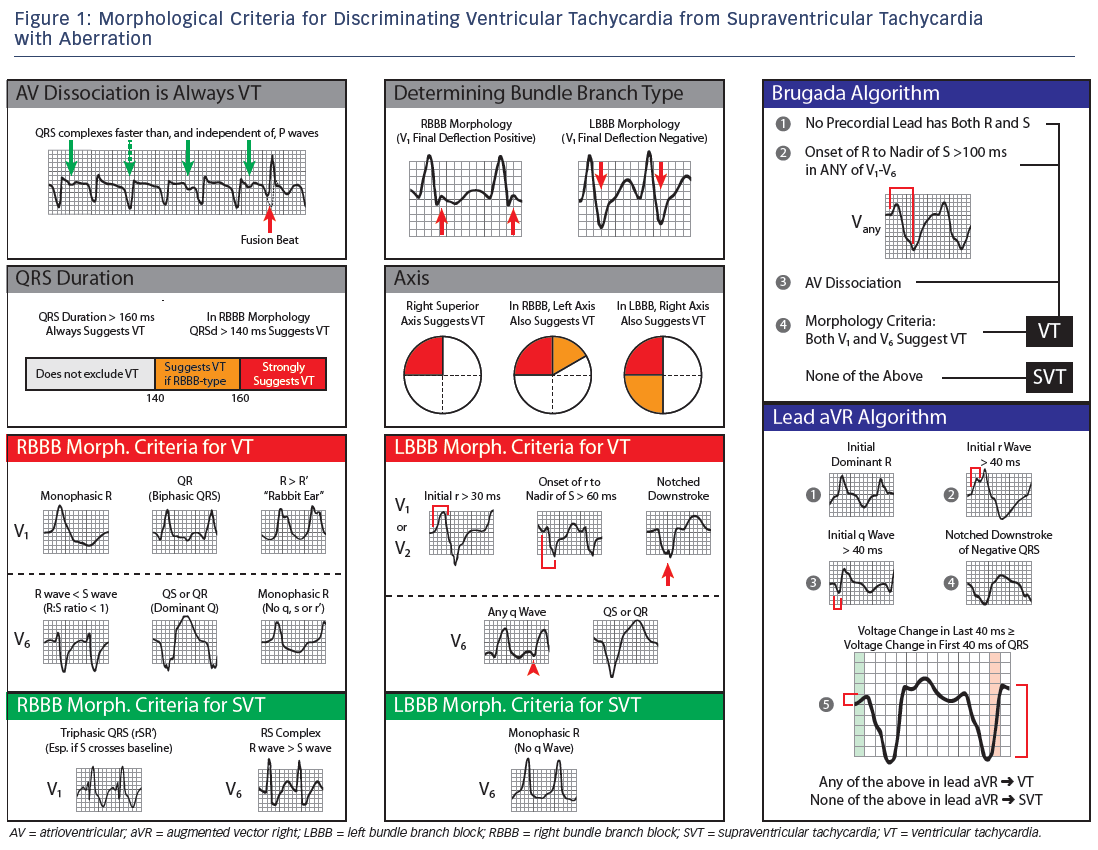
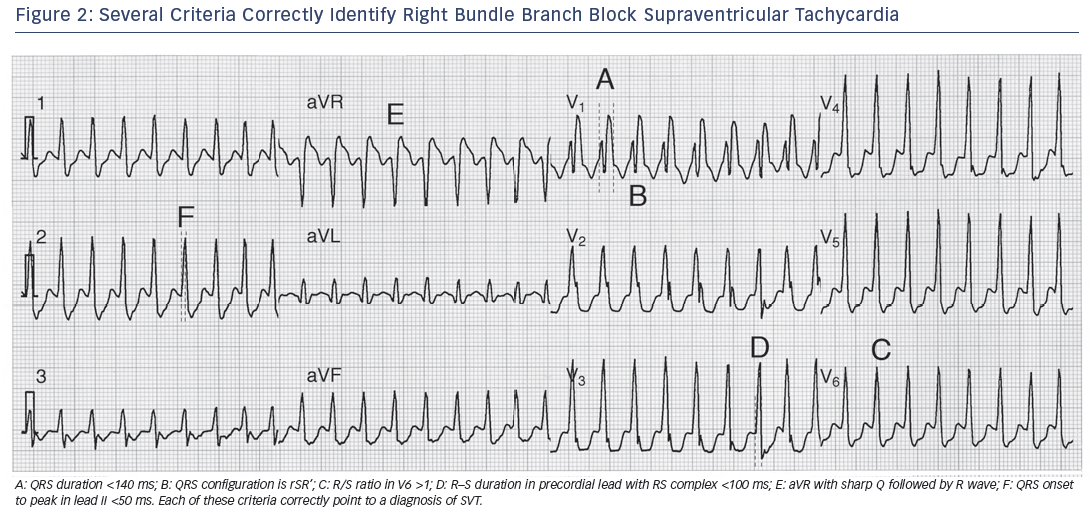
Specific Ventricular Tachycardia Algorithms and Their Performance
In Table 2, we list many of the most popular VT criteria. Where a criterion is to be applied only to one BBB pattern or another, this is listed before the criterion as [RB] or [LB] for RBBB and LBBB, respectively. Many of these criteria (e.g. AV dissociation = VT) suggest one diagnosis, but do not exclude the opposite diagnosis. For such criteria, a predictive value is listed. For algorithms that can give both SVT and VT as possible answers, we report overall accuracies. In all cases, these numbers were stated explicitly in the original reports or derived from them using standard formulas and 2x2 tables.
Foundations – The Morphology Criteria
The work of Sandler and Marriott published in 1965 laid the foundation for the use of ECG criteria instead of, or in complement to, physical exam skills for the diagnosis of VT. Their pioneering work showed that conventional assumptions about how normal ECG patterns ‘should’ present were often misleading. From analysis of 100 premature ventricular contractions (PVCs), 50 RBBB aberrancies and 100 fixed RBBBs, they drew many conclusions, a few of which have withstood the test of time. The generally accepted morphology criteria from their work as well as that of other investigators are summarised in Figure 1.
Sandler and Marriott Criteria (1965)
- [RB] Identical activation vector = SVT. If the initial 20 ms of the QRS are the same in WCT as in sinus rhythm, SVT is favoured by 20:1, with a positive predictive value (PPV) of 92 %.6 Of course, the sinus rhythm ECG must be available for this analysis.
- [RB] An rSR’ where S crosses baseline = SVT. The presence of such a QRS in a RBBB WCT favours SVT by at least 11:1 with a PPV of 91 %.6
- [RB] Triphasic QRS = SVT. A triphasic QRS in V1 favoured SVT with a PPV of 92 %.6
- This criterion’s distinguishing power performs well even in studies designed to minimise the performance of VT criteria, with specificity ≥90 %.7
- [RB, LB] Precordial concordance = VT. A QRS, which is predominantly positive or predominantly negative in every precordial lead, overwhelmingly favours VT.8 Subsequent studies have confirmed this with specificity of 95–100 % and a PPV of 89–100 %.7,9,10
The Wellens Criteria of Right Bundle Branch Block
By 1978, two advances had allowed a leap forward in the ECG criteria: the development of the His bundle electrogram and simultaneous multi-lead electrocardiography. Using these new tools, Wellens et al. analysed 70 sustained VT and 70 SVT episodes with aberrancy, all proven at electrophysiological study.11 Though the criteria set forth in this paper are often discussed only in terms of their contributions to discriminating RBBB tachycardias, this paper also offered observations on LBBB morphology that would be used by later authors. As they are commonly used today, Wellens’ criteria for RBBB VT are as follows:
- [RB] QRS duration >140 ms = VT. The original data showed a specificity and PPV of 100 % for VT. Subsequent studies found this less certain, with specificities of 57–75 % and PPV of 89 %.7,10
- [RB] Left axis = VT. This was originally discussed without regard to bundle block morphology, but it is most robust for RBBB WCT where the original PPV was 94 %. Later studies have found PPVs of 88–94 %.9,10 With extreme left axis (more negative than -90 °), the PPV is 98 %.
- AV dissociation = VT. Of all criteria, this is the most secure. Six separate, large cohorts have all found 100 % specificity and 100 % PPV for true AV dissociation as a marker of VT.9–13 It holds true regardless of bundle branch pattern or other morphology criteria. Its weakness is being able to confidently recognise its presence; in many cases, even when AV dissociation is clearly present on intracardiac recordings during VT, it cannot be readily seen on the ECG.
- [RB] Morphology criteria. Wellens built on the observation of Sandler and Mariott that mono- or biphasic V1 QRS morphologies in a RBBB WCT suggests VT. Though the original paper found a 97 % PPV for this statement, later study has been unable to confirm this; finding a PPV of only 82–83 %.7,9,12 If the V1 QRS is triphasic, Wellens’ criteria suggests investigation of V6 for an R:S ratio <1 (that is, R wave smaller than S wave) as suggesting VT. This criterion had a modest 90 % PPV both in the original data and subsequent evaluation.10
- [RB] ‘Rabbit ears’ Rsr’ = VT. Aside from the four well-known criteria above, Wellens noted that an unusual triphasic V1, with the left R wave taller than the right, and the S wave not crossing the baseline, was invariably associated with VT; subsequent study has confirmed this 100 % PPV.9
The Kindwall Criteria of Left Bundle Branch Block
Building on the observations of V6 behaviour in LBBB noted by Wellens and others, Kindwall and Josephson published the first and still most common criteria specific to LBBB WCT.14 Each of the criteria alone has poor sensitivity but high specificity (and thus high PPV, ≥97 %). To remedy this, the criteria are applied such that presence of any of the four criteria indicates VT, with an overall accuracy of 97.5 % in the original study and excellent performance in subsequent literature.9
These criteria are:
- [LB] V1 or V2 with initial R >30 ms = VT.
- [LB] V1 or V2 QRS onset to nadir of S wave >60 ms = VT.
- [LB] V1 or V2 with notching on the S wave downstroke = VT.
- [LB] Any Q in V6 = VT.
The Brugada Criteria
Published in 1991, the Brugada criteria were the first to offer applicability to all WCT without limitation to one BBB configuration or another. The original paper reported overall accuracy of 98 %. However, no subsequent study has been able to achieve such results, with the overall accuracy of the algorithm 77–85 % across four large studies.13,15–17 Most authors note difficulty in applying the last step of the criteria (the morphology section), particularly among noncardiologists. The criteria are applied in stepwise fashion, stopping further analysis if any step suggests VT.
- Step 1: Absence of RS complex anywhere in V1–V6 = VT.
- Step 2: Onset of R to nadir of S in any precordial lead >100 ms = VT.
- Step 3: AV dissociation = VT.
- Step 4: Morphology criteria (see Figure 1 for details). Of particular note, both V1 and V6 must suggest VT for the diagnosis to be made; otherwise, SVT is the diagnosis.
Recognising that the morphology criteria can be difficult to remember, some advocate using only steps 1 and 2. The results of this approach have been variable with PPV for VT of 81–96 % in two different studies.10,17
The Vereckei Criteria of Augmented Vector Right
Vereckei et al. were the first to suggest the possibility of diagnosis of VT in either left or right BBB WCT from a single lead – augmented vector right (aVR) in this case.18 In similar fashion to the Brugada criteria, this algorithm is applied stepwise, stopping if VT is ever suggested, and ending with SVT if no criteria suggests VT. The original algorithm was modified in a subsequent paper for increased ease of use13 though other authors have noted that the application of the ventricular activation velocity ratio (Vi/Vt) criteria (needed in up to 50 % of cases) can be frustrating and imprecise at standard ECG scales and paper speeds.16 Thus, the accuracy of the algorithm, 92 % in the original paper, was far less in a subsequent large study (72 %). The Vereckei (2008) criteria are:
- Step 1: An initial, dominant R in aVR = VT.
- Step 2: An initial, non-dominant q or r in aVR >40 ms = VT.
- Step 3: Notching on an initial downstroke in aVR = VT.
- Step 4: Vt≥Vi in aVR = VT. To apply, measure the total vertical distance covered by the final 40 ms of the QRS in aVR. If this is equal to or more than the vertical distance covered by the first 40 ms of the aVR QRS, VT is diagnosed. The concept is that with aberration, ventricular activation during the first portion of the QRS is mediated by the His-Purkinje system, whereas in VT, the His-Purkinje system is engaged later in the QRS complex.
The Pava Criteria of Lead II
In 2010, Pava et al. published the second algorithm offering diagnosis from a single lead, without regard to BBB morphology, and this time using only a single relatively simple measurement.19
This is the R wave peak time in lead II, with the interval from QRS onset to first change in polarity (R or S peak) in lead 2 ≥50 ms denoting VT.
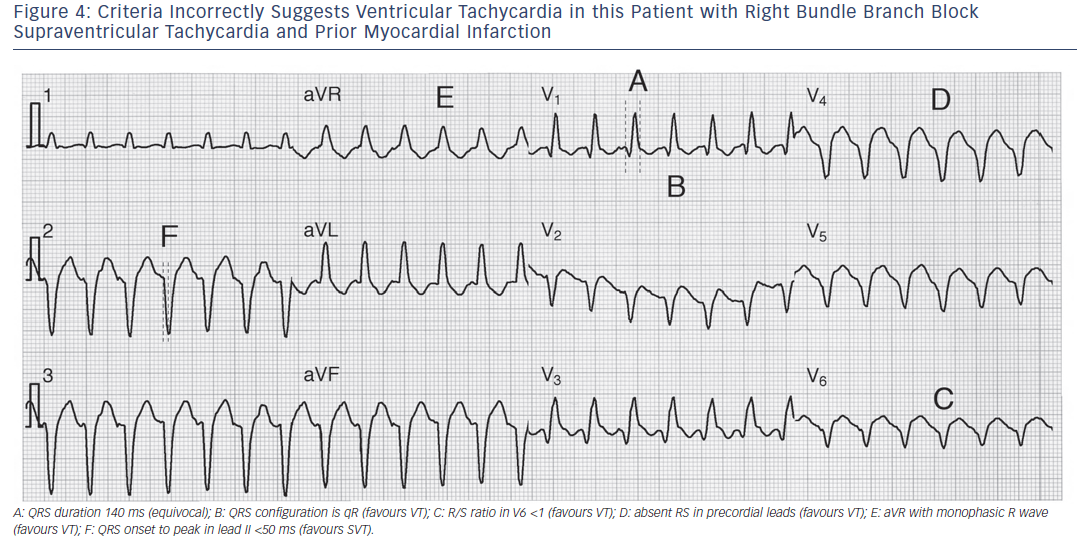
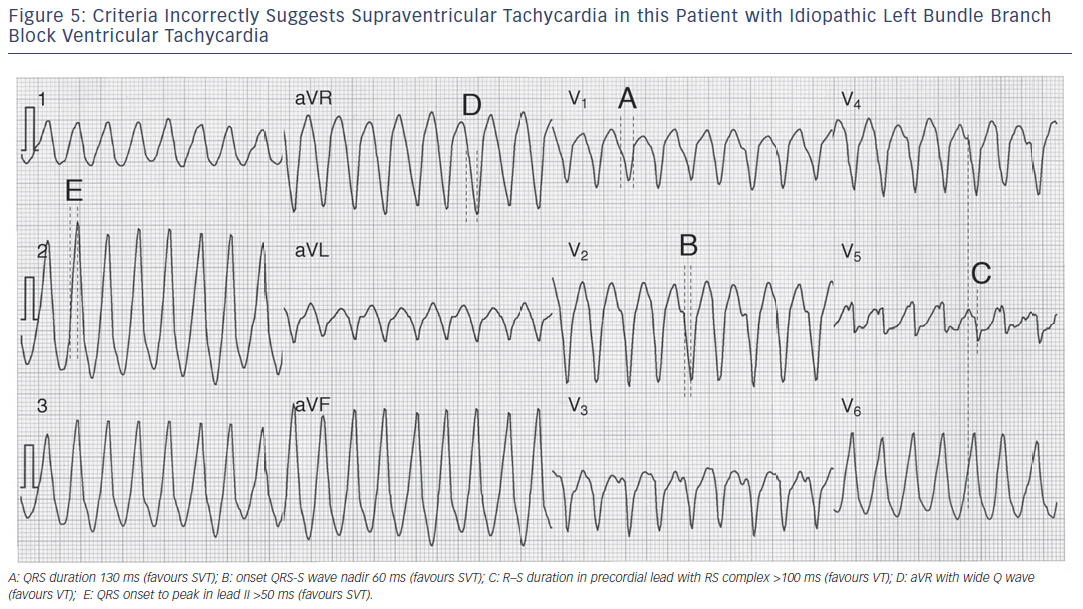
The original paper reported remarkable test characteristics with an area under receiver operator curve exceeding 98 %, specificity of 99 % and PPV of 98 %, meaning the algorithm promised to distinguish VT from SVT with 94.5 % accuracy from a single measurement. The algorithm’s performance was substantially less favourable in its first large external application, with an overall accuracy of just 69.0 % compared to, for example, the Brugada criteria (77.5 % accurate) in that same study.16 Further study is needed to determine the true value of this criterion. Application of many of the foregoing criteria is shown in Figures 2-5).
A Different Approach – The Griffith Algorithm
Recognising the high prevalence of VT and the limited number of typical aberrancies allowed by the conduction system, Griffith et al. proposed criteria, which function in a manner counter to the algorithms above – each ECG is analysed for V1 and V6 criteria consistent with aberration.20 If the criteria for aberration are not found, VT is assumed. As the algorithm defaults to VT, its sensitivity can be excellent, but specificity (and therefore overall accuracy as well) suffer, both in the original paper and in subsequent evaluation.16,17,20 Of note, right ventricular outflow tract tachycardias are frequently misclassified as SVT by this algorithm, and should be evaluated with other criteria by users of this algorithm.
Embracing the Unknown – Bayesian Analysis
A Bayesian algorithm has been validated, which recognises that a definitive answer is not always possible for any given criteria. This unique algorithm relies on likelihood ratios and a pre-test odds ratio of 4 (representing the 80+ % chance a given WCT will be VT) to compute a final likelihood of VT from any number of its component criteria, and readily lends itself to the addition of new criteria as they emerge, though these would require validation in another cohort.9
Specific Noteworthy Ventricular Tachycardias
Three types of VT (right or left ventricular outflow tract VT, and fascicular VT) are particularly amenable to treatment by drugs, catheter ablation or both. These largely benign forms of VT are particularly important; their presence, in the setting of a structurally normal heart, is a contraindication for ICD implantation.21 Diagnosis of these forms of VT is more difficult than the VT-related to structural heart disease, because the QRS duration is comparatively shorter, VA conduction is often present (not dissociated), and many other criteria fail to distinguish these VTs from SVT (as in Figure 4). An additional VT-type, bundle branch re-entry (BBR), usually has an appearance indistinguishable from LBBB SVT (propagation anterogradely over RBB, retrogradely over LBB) and thus belongs in the general category of VTs that are difficult (if not impossible) to distinguish from SVT. While BBR VT is also quite amenable to catheter ablation, it generally occurs in patients with significant structural heart disease (cardiomyopathy); thus most patients with BBR VT also warrant ICD therapy.
Remaining Problems
Though published criteria to diagnose WCTs demonstrate admirable test characteristics in their initial reports, clinicians still have difficulty arriving at the correct diagnosis. Potential causes for this are many, including:
- complexity of differentiation criteria;
- unfamiliarity with the criteria;
- incorrect application of criteria (i.e. misreading a QRS duration); and
- disbelieving the results of applying the criteria (“I know everything points to VT, but he looks so good that it must be SVT”).
Certainly, the very simplified criteria set forth by Vereckei and Pava in recent years go a long way toward rectifying the first two obstacles to correct diagnosis, but remedies for the other shortcomings may not be as readily forthcoming.
Conclusion
The diagnosis of VT has undergone evolution and development in concert with the field of cardiology itself, but the necessity of a correct diagnosis remains unchanged. The world has not yet seen the ‘one criterion to end all criteria’, and it seems unlikely to appear in our near future. Until that time, maintaining familiarity with several of the available criteria will assist any provider in delivering the proper care when it is needed most. When all else fails, it is wisest to treat the patient initially as though the diagnosis was VT (which, as noted, is correct about 80 % of the time) and leave the fine-tuning of diagnosis and long-term management plan for later.








