Catheter ablation of AF requires ablation of the posterior wall in the left atrium. With conventional thermal ablation modalities, the lesion can spread past the myocardium to affect adjoining structures, which is a common source of certain AF complications. One such structure is the oesophagus, which is immediately adjacent to the left atrial posterior wall.1 Oesophageal damage can manifest as asymptomatic lesions that can be seen only with imaging; occasionally symptoms such as pain and dysphagia are present. The most feared of all AF complications is the development of the mostly fatal atrio-oesophageal fistula. Accordingly, different strategies have been employed to protect the oesophagus from the risk of thermal damage. These include modifications of energy delivery such as high-power short-duration ablation, luminal oesophageal temperature monitoring, mechanical deviation of the oesophagus away from the thermal source or direct cooling of the oesophagus during radiofrequency ablation (RFA).2
Pulsed field ablation (PFA) is a novel energy source for catheter ablation, which uses electrical fields to mediate myocardial death via irreversible electroporation (IRE) as well as other mechanisms. Different cell types have varying thresholds for IRE, thus PFA promises to potentially reduce collateral damage based on its tissue specificity. The combination of a non-thermal, tissue-specific ablation has sparked enthusiasm for PFA, and several early preclinical and clinical reports now support these attributes.3,4 However, certain complications such as coronary artery spasm during mitral isthmus ablation have only been observed after the clinical implementation of left-atrial PFA in Europe.5 Thus, it is imperative to stay vigilant about potential complications – even if deemed unlikely – when dealing with novel ablation energies and catheters. Although rarely, atrio-oesophageal fistulas continue to occur with thermal ablation modalities.6,7 Given its exceedingly high mortality, this complication is particularly important to evaluate for PFA. The aim of this article is to review the effects of PFA on the oesophagus and to attempt to address the risk of severe forms of oesophageal injury.
Literature Search and Output
To identify studies evaluating the effects of PFA on the oesophagus, a databank search (PubMed, Ovid, Google Scholar, Web of Science, Scopus) was performed using the keywords ‘pulsed field ablation’ OR ‘electroporation’ AND ‘esophagus’. Only studies that presented original data were included. Preclinical studies were only considered if histology was used for the assessment of the oesophagus. In total, 18 studies were included in this literature review. Nine preclinical studies used a porcine animal model with 57 animals in total, and one preclinical study used a rabbit model with 36 animals. An overview of the porcine animal studies is displayed in Table 1. Eight studies evaluated the effects of PFA in the clinical setting and included 2,438 patients in total. Clinical studies are summarised in Table 2.
Preclinical Data
Preclinical studies for novel ablation systems are performed to assess feasibility, to develop dosing strategies and for safety assessments. This is imperative as they can help understand lesion behaviour as predicted by modelling studies. We can also assess supratherapeutic energy delivery in a ‘worst-case’ scenario – an approach that allows evaluation of the potential of the catheter and the energy source for myocardial and collateral tissue damage that may not be realised during standard preclinical testing and early clinical use. The ability to assess the organ at risk using histology facilitates the detection of even minor subclinical damage and can help prevent serious complications from occurring in patients. However, one should also be aware of the limitations of preclinical studies: findings from these studies cannot be directly translated to humans, especially as they cannot recapitulate the wide variations in ablation approaches used by clinicians and cannot account for underlying disease states that may predispose to complications. The comparatively small nature of preclinical studies reduces the likelihood of capturing infrequent complications that may only manifest once the technology is widely used.
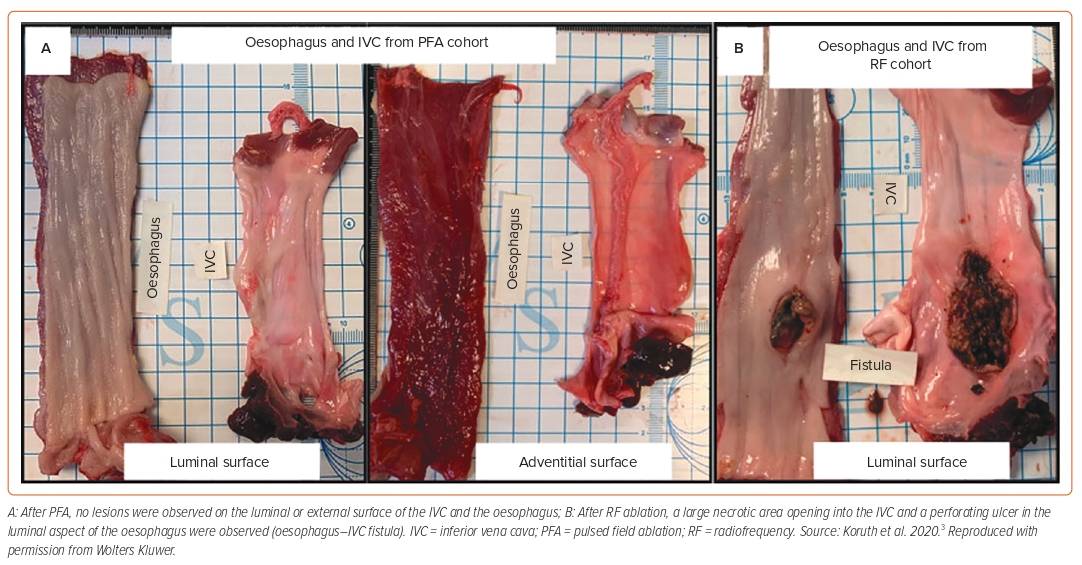
Several models have been used to evaluate the potential of PFA systems to damage the oesophagus. One approach is to expose the oesophagus to supratherapeutic pulsed fields by using a balloon device within the oesophagus to forcefully deviate it towards the ablation catheter placed within the inferior vena cava (IVC) or the right atrium.3,8–11 With this model, control animals treated with RFA consistently showed severe oesophageal damage, proving the utility of the model (Figure 1). 3,8,9 Other approaches used to deliver supratherapeutic PFA to the oesophagus include ablation from within the aorta towards the oesophagus, or from within the cardiac chambers close to the oesophagus, but without deviation.12–14
The published reports employing these assessments for PFA technologies all had a survival period of 14–30 days; none reported any oesophageal damage even on histology after PFA.3,8–14 Two additional preliminary reports using the deviation model described the presence of oesophageal lesions secondary to PFA exposure; however, they differ from the above reports in that the oesophagus was examined at the acute time point (short-term survival).15,16 One of these reports examined the oesophagus at 2 hours and the other at 48 hours post-ablation. Both reports used PFA systems different from the other reports but importantly demonstrated that, despite acute lesions, no lesions were seen when the animals survived chronically.15,16
Three separate preclinical reports evaluated the effect of PFA on the oesophagus by direct application of PFA on (open surgical) or from within the oesophagus.11,17,18 Of note, all three reports included oesophageal assessments shortly (1–2 days) after ablation. Neven et al. used a custom ablation device applied directly on the surgically exposed oesophagus in eight swine.17 The authors observed small white densities on endoscopy after 2 days, which had completely resolved by the seventh day of follow-up.17 Yavin et al. introduced a lattice-tip catheter capable of endocardial PFA delivery into the oesophageal lumen and delivered PFA.11 The authors reported mild, superficial oedema and inflammation, along with focal necrosis of the oesophageal wall in these animals.11
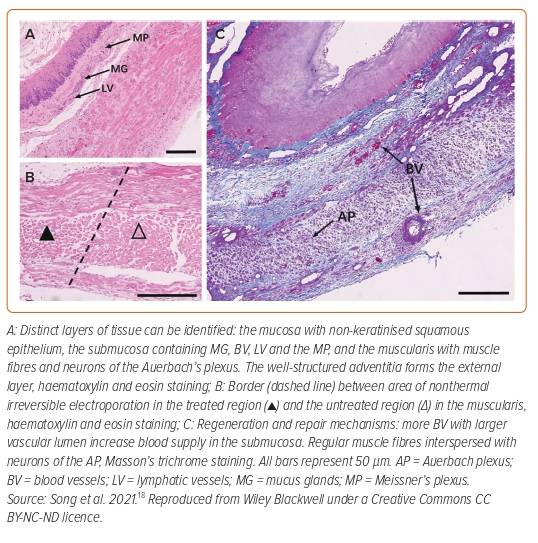
Finally, Song et al. performed PFA on 84 New Zealand rabbits to assess the acute and long-term effects of PFA on the oesophagus.18 Forty-eight animals were used for dosing assessments and on the remaining 36 animals, monopolar PFA (70 µs pulse duration, nine trains with 10 pulses each, 2.5 kV/cm) was directly applied to the oesophagus via two plate electrodes. Animals were divided into six groups of six animals each. One animal in each group underwent a sham procedure without ablation and served as a control. After 1 and 3 days and after 1, 2, 4 and 16 weeks, animals were sacrificed, and histology was performed. The study design allowed for comprehensive assessment spanning both the acute as well as chronic phases of post-PFA oesophageal effects. Only seven of 36 animals developed mild symptoms of anorexia after ablation, which resolved spontaneously within 3 days. Acutely (days 1–3) after ablation, evidence of lesion formation with inflammatory reaction across the wall was observed. There was significant oedema of the submucosa, and the muscular layer was noted to be completely ablated. There was no haemorrhage noted and blood vessels (arterioles, veins) were spared. After 3 days, the general architecture of the oesophageal wall (mucosa, submucosa, muscularis and adventitia) remained intact, with the extracellular matrix noted to be spared. In the groups that survived for 2–4 weeks, lesions had almost completely healed, with oesophageal wall constituents, such as plexuses, mucus glands and muscle fibres (Figure 2), being easily visualised with normal morphology.18 The authors concluded that after PFA near the oesophagus in rabbits, conditions for complete tissue repair were preserved and, therefore, that severe complications, such as fistula or perforation, are unlikely.18
In summary, preclinical data demonstrate that PFA when applied directly atop the oesophagus – a worst-case scenario that exaggerates the effects of endocardial delivery performed in patients – can induce acute lesions that can be transmural, but which then heal without evidence of permanent damage over 1–2 weeks. In other studies using focal or multielectrode catheters designed for clinical use, when PFA was delivered within the blood pool but close to the oesophagus, no oesophageal lesions were observed chronically. However, preliminary reports with these catheters and shorter survival periods suggest that these PFA systems can create acute, non-transmural oesophageal lesions.
Clinical Data
Clinical studies provide additional confirmation of the insights gained from preclinical data. The large numbers of patients included in clinical studies facilitate the detection of rare complications that may not have occurred in the smaller preclinical assessments, but nevertheless remain limited as certain complications require several thousand patients to manifest. The main limitation of current clinical studies of left atrial PFA is the aggressiveness with which oesophageal involvement has been investigated. Histology is not feasible in clinical studies, so subclinical lesions can be very challenging to detect. As mucosal lesions are manifestations of transmural oesophageal lesions, thus representing the most severe form of injury, endoscopic assessment for PFA effects (the most frequently performed investigation) is, by definition, not very sensitive. However, certain clinical studies have employed different diagnostic tools to evaluate oesophageal reactions to PFA. Cochet et al. systematically performed cardiac MRI acutely (<3 hours) and 3 months after pulmonary vein isolation (PVI) in 41 patients.4 Of 24 patients treated with thermal ablation modalities, direct contact between the oesophagus and an atrial ablation site was observed in 11 (46%). Ten (91%) of these patients showed late gadolinium enhancement as a sign of oesophageal damage on the acute MRI. No signs of oesophageal involvement were observed in the 18 patients treated with PFA, despite 11 (61%) of these patients demonstrating contact between the oesophagus and an ablation site. Repeat MRIs 3 months post ablation showed complete resolution of all injury. In a subsequent evaluation, Reddy et al. performed MRI post ablation in 18 patients and also reported no signs of oesophageal damage.19 In several other studies and registries, endoscopy was used to assess oesophageal damage post ablation.19–23 In total, 2,438 patients were included, of whom 211 (9%) underwent endoscopy within the first week post ablation. A total of 2,136 (88%) of patients were treated with the pentaspline catheter (FARAWAVE, Boston Scientific), while a variable-loop circular catheter (Biosense Webster) and a lattice-tip catheter (Sphere-9; Affera, Medtronic) were used in 226 (9%) and 76 (3%) of patients, respectively (Figure 3). Additional posterior wall ablation was performed in 206 (8%) of patients. By far the largest group of patients was included in the MANIFEST-PF registry.24,25 This multicentre study recruited 1,758 patients treated with PVI using the pentaspline catheter. None of the studies published to date have reported any oesophageal complications or abnormal endoscopic findings after the clinical use of PFA.
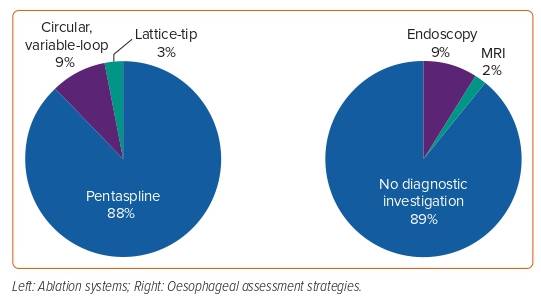
The designs of clinical studies published thus far have been heterogeneous. Several prospective studies have systematically assessed the oesophagus in all or most patients after ablation.4,20,21 However, these studies are limited by the comparatively small number of patients included. In the studies with cohorts of >100 patients, only a smaller subset (18–31%) of patients received endoscopy post ablation.19,22,23 Duytschaever et al. performed endoscopy only in the 40 patients who were included in the first study phase for safety assessment.23 In the two remaining studies, it was not reported how patients were selected for endoscopy.19,22 In the MANIFEST-PF registry, no data on post-procedural endoscopy were reported.24,25 Only two trials used oesophageal temperature monitoring, one of which reported “low-level oesophageal heating (typically <39°C)” during ablation of the posterior LA-wall.21,22 An overview of all clinical studies analysed for this review is provided in Table 2.
Discussion
The current assessment of PFA and risk of oesophageal damage can be summarised as follows:
- Preclinical reports suggest a range of effects, from no effect (albeit based only on chronic assessment at which point acute lesions may have resolved) to that of acute oesophageal lesions.
- Acute PFA lesions observed preclinically within ≤2 days post ablation can range from mild to severe transmural lesions. The severe lesions were seen in the setting of supratherapeutic PFA delivery specific to the preclinical model.
- All such lesions demonstrated near complete healing by 1-2 weeks post ablation.
- No relevant oesophagus damage has been reported clinically thus far.
Before we discuss the observations derived from the several reports mentioned above, it is important to acknowledge both the complexity of PFA waveforms and the lack of transparency of PFA dosing by manufacturers of systems used today for cardiac ablation. Currently, several ablation catheters exist, some with a focal design (ranging from 3.5 mm to 9 mm tip size) that can indent tissue to a greater degree than larger footprint catheters designed for ‘one-shot’ PVI. In addition, there are several waveform/repetition and dosing differences between systems. All these factors, superimposed on clinical disease states in patients that cannot be captured in animal models, and off-label use of these systems, limit our ability to predict the risk of oesophageal injury.
The acute and sub-acute effects of PFA on the oesophagus can be gleaned from preclinical studies in which the oesophagus was assessed acutely post ablation. Of note, these studies reported effects of PFA on the oesophagus, with either direct-atop the oesophagus ablation or after forceful deviation of the oesophagus (both of which do not reflect the nature of PFA exposure seen in clinical left atrial ablation).11,15–18 These reports also indicate that oesophageal lesions healed chronically without leaving signs of damage, likely because the oesophageal extracellular matrix was preserved and acted as a scaffolding for repair mechanisms.18 This preservation of the extracellular matrix, believed to be universal to all forms of PFA, is critical to the belief that, even if lesions do occur with all or certain PFA systems, fistula formation will not ensue. Given the lack of consistent acute and chronic testing with all catheters and waveforms it is difficult to state whether waveforms exist that absolutely spare the oesophagus.
Nevertheless, the clinical data published so far are promising. Even with several thousand patients included in the studies mentioned above, there have been no reports of oesophageal damage. This is true for studies that only used clinical follow-up as well as for those that implemented a specific strategy to investigate oesophageal involvement. The results suggest that, in a clinical setting, PFA likely spares the oesophagus. A recent article reviewing clinical studies about PFA arrived at the same conclusion.26 However, several limitations regarding the data need to be considered, and we likely underestimate the full extent of oesophageal involvement. Most patients (88%) this far have been treated with a single ablation system featuring a pentaspline catheter. As every system uses its own, proprietary waveform, the apparent safety regarding the oesophagus may not be generalisable to other systems. Therefore, continued sensitive safety assessments should be repeated carefully for current and future PFA systems. Endoscopy was the method most commonly used for the assessment of oesophageal damage. However, this diagnostic tool can only detect macroscopic, transmural lesions. MRI and other modalities can be more sensitive to detecting subclinical involvement of the oesophagus and should be pursued for all technologies. Also, repeat ablation procedures using PFA might have a different risk profile than that of index ablations. More studies are necessary to evaluate the risk of repeat PFA procedures, especially after index ablation with thermal ablation modalities.
Conclusion
Preclinical studies have shown that supratherapeutic PFA can induce acute oesophagus lesions. Several features of these PFA-induced lesions suggest that such lesions are likely not going to lead to transmural perforation and are therefore even less likely to induce atrio-oesophageal fistula formation. However, preclinical healing patterns may not be replicated in patients, and it may take upwards of several thousands of patients including those who undergo ablation of persistent AF (where multiple repeat applications may be performed) for us to truly conclude that fistula formation is a thing of the past. 
Clinical Perspective
- There are no reports of adverse events regarding the oesophagus after endocardial pulsed field ablation in the clinical arena thus far.
- However, based on the preclinical evidence that oesophageal lesions can occur with certain waveforms, further clinical testing with sensitive imaging modalities is needed.
- Based on the preservation of tissue scaffolding with pulsed field ablation and the consistent pattern of healing seen in preclinical testing, it appears that the risk of severe oesophageal complications such as fistula is likely very low to none with current generation pulsed field ablation catheters.