Monitoring after implant of patients with cardiac implantable electronic devices (CIED) forms a part of both device and patient care, and is the responsibility of the implanting centre. Monitoring is challenged by the increasing number and technical complexity of implanted devices coupled with the increasing clinical complexity of the patient population. Current practice is based on quarterly to yearly in-office visits with an increased rate when the device approaches its end of service, or in case of advisories.1–3 In this model, download of data stored in the device memory, potentially useful for patient management, is delayed. Telemedicine offers a unique opportunity to optimise clinic workflow and to improve device monitoring and patient management.
Technology
All major CIED manufacturers have introduced remote monitoring (RM) systems.4 All of them are based on a patient unit capable of interrogating the device and downloading the programmed parameters and the diagnostic data. Information is transmitted to a central database where it is decrypted and stored on a secure website on which it can be viewed by the clinical staff. Early wand-based systems required patient-driven downloads relayed via telephone connections to following facilities. Currently available systems are based on automatic transmission mechanisms that are fully independent of patient or physician interaction. A distinction has to be made between ‘remote interrogation’ and ‘remote monitoring’. In the first, device interrogation is performed periodically at home either by the patient manually or by the monitoring system automatically at predefined intervals. In the second, continuous device monitoring may trigger unplanned transmissions in case of programmable alerts. In spite of major commonalities, different proprietary systems may differ substantially in connectivity, patient involvement, transmission scheduling and alert availability and programming.
Impact on Out-patient Clinic Workload
The Lumos-T Safely Reduces Routine Office Device Follow-up Trial (TRUST)5 was the first large randomised trial which demonstrated that RM may safely reduce the number of in-hospital visits by nearly 50 %. The reduction primarily occurred in scheduled encounters involving collection of routine measurements and requiring no clinical intervention. Unscheduled visits slightly increased in the remote arm. The overall reduction in face-to-face visits was obtained safely, with no difference between the two study arms in mortality, incidence of strokes and events requiring surgical interventions. Furthermore, detection of arrhythmia onset was anticipated by more than 30 days. Regarding patient adherence to follow-up, the TRUST results6 showed that patients in the RM arm more effectively and durably attained follow-up goals of punctual scheduled follow-up and patient retention compared with conventional methods. Several studies have confirmed such results and there is now strong evidence that RM significantly reduces in-hospital visit numbers, time required for patient follow-up, physician and nurse time and hospital costs.7–11 Furthermore, social costs for patients and caregivers which include travelling, missed work and social activities may be reduced by RM.12,13Device Management
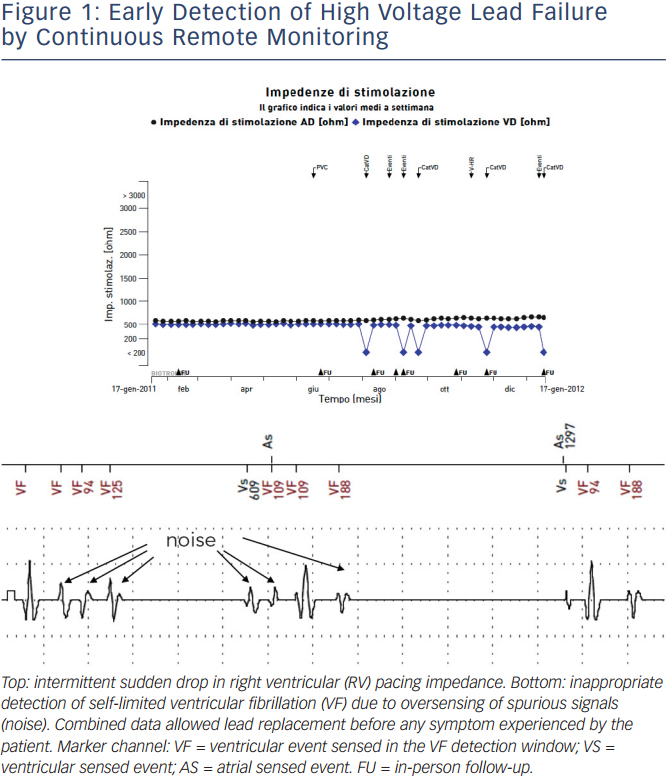
Lead and device performance monitoring is a physician responsibility and represents a challenge due to the number and complexity of implanted systems and the increased number of advisories during the last 10 years. A strategy based on frequent in-hospital follow-up visits is unsuitable as it places a huge work burden on out-patient clinics. It is also inefficient since malfunctions are rare and likely to be missed when developing suddenly in the interval between visits, with the possibility of causing potentially life-threatening complications.14 RM allows continuous monitoring, with automatic alerts of battery voltage and impedance, circuit status, charging time, low- and highvoltage lead impedances, sensing and pacing threshold values, external interferences, inappropriate detection of arrhythmias due to noise and double counting or T-wave oversensing (see Figure 1). Clinical studies have demonstrated that malfunctions are usually asymptomatic and that RM is superior to a standard strategy for early detection.15–18 Impact of RM on device longevity is a matter of debate. Concerns have been raised about the potential negative impact of monitoring itself on battery drain. On the other hand, continuous monitoring may allow device programming optimisation with reduction of battery drain. Reduction of capacitor charges by 75 % in implantable cardioverter defibrillator (ICD) patients randomised to RM has been demonstrated in the Effectiveness and Cost of ICD Follow-up Schedule with Telecardiology (ECOST) trial with a potential major impact on battery longevity.19
Disease Management
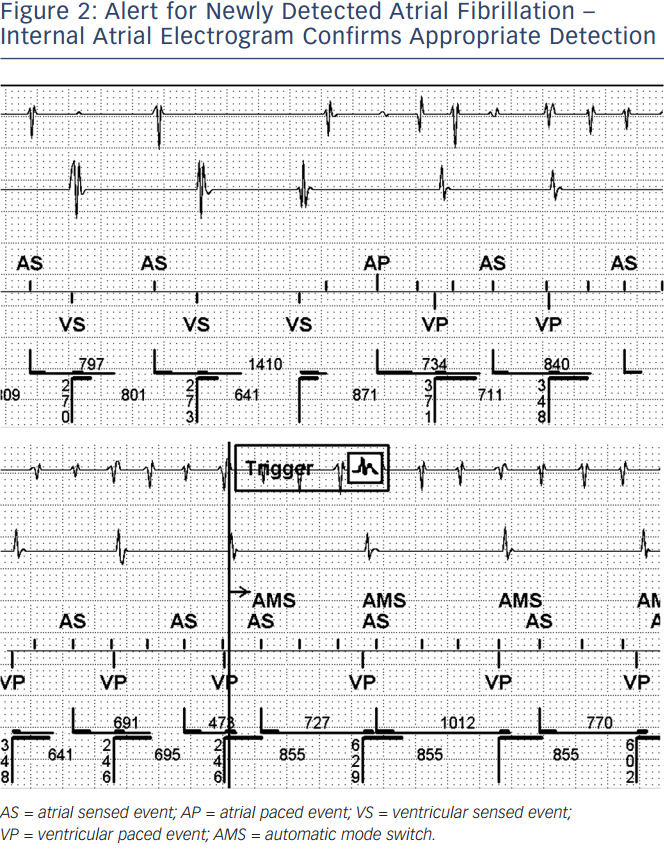
Atrial Fibrillation
Expected benefits of RM in patients with CIED and atrial fibrillation (AF) are mainly represented by early arrhythmia detection and patient continuous monitoring. Early detection of AF may induce prompt clinical reaction aimed at preventing severe adverse events such as stroke and heart failure.20 Continuous monitoring allows individual tailoring of patient treatment and continuous updating of therapeutic strategy. AF is very common in CIED patients even in those without any history before implant. Furthermore the majority of events are asymptomatic.21 CIEDs keep detailed information about AF episodes, including number and duration, arrhythmia recurrences and burden, mean and maximum ventricular rate and intracardiac electrogram strips (see Figure 2).22 RM allows continuous access to stored data, and alerts may be programmed for specific events. Early detection of AF by RM has been demonstrated by several trials (for instance five days versus 31 days in the TRUST trial).5 Clinical evidence for stroke risk reduction by RM is still awaited. Preliminary studies estimated that daily monitoring may reduce the two-year stroke risk by 9–18 % with an absolute reduction of 0.2–0.6 %, compared with conventional inter-visit intervals of 6–12 months.23,24 In the Comparative Followup Schedule with Home Monitoring (COMPAS) trial, stroke rate was significantly higher in the control group than in the RM group (3.3 % vs 0.8 %).8 In the recent Anticoagulation Guided by Remote Rhythm Monitoring in Patients With Implanted Cardioverter-Defibrillator and Resynchronisation Devices (IMPACT) study25 of oral anticoagulation therapy for AF guided by RM (started in the case of AF detection, stopped in the case of no recurrences), there was no difference in the outcomes of stroke or all-cause mortality between the intervention group and controls. As a matter of fact, 92 % of patients who experienced stroke in the study group were either not anticoagulated at all or had an international normalised ratio (INR) < 2 at the moment of the event.
Download origina
Considering that several trials have demonstrated no temporal relationship between AF episodes and stroke it should be recommended that, once started for AF, anticoagulation is not discontinued in the absence of device-detected AF.26,27
VentricularArrhythmias
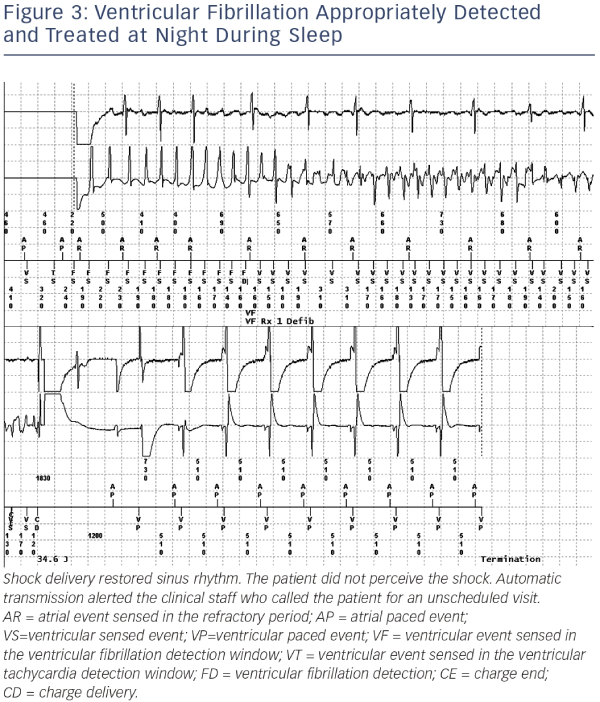
The significant advantage of RM is the prompt evaluation of appropriateness of detection and efficacy of therapy delivered (see Figure 3). Wireless devices send a virtually immediate transmission for review. The physician can evaluate the episode detail on the website, including internal electrograms and marker chain. With inductive systems, patients may manually send a transmission to the service centre and inform the referring physician by phone in case of perceived shock and/or palpitations or syncope. Ability of RM to early detect ventricular tachyarrhythmias has been demonstrated by the TRUST trial5 (one day vs 36 days for ventricular fibrillation and one day vs 28 days for ventricular tachycardia). Another potential benefit of RM is prevention of inappropriate shocks and also of appropriate but unnecessary shocks. Early reprogramming after inappropriate detection and therapy modulation in the case of slow, well-tolerated arrhythmias may represent the mechanisms for reaching this goal. The ECOST trial demonstrated that RM significantly reduced the number of actual delivered shocks (-72 %), the number of charged shocks (-76 %) and the rate of inappropriate shocks (-52 %).19,28
Heart Failure
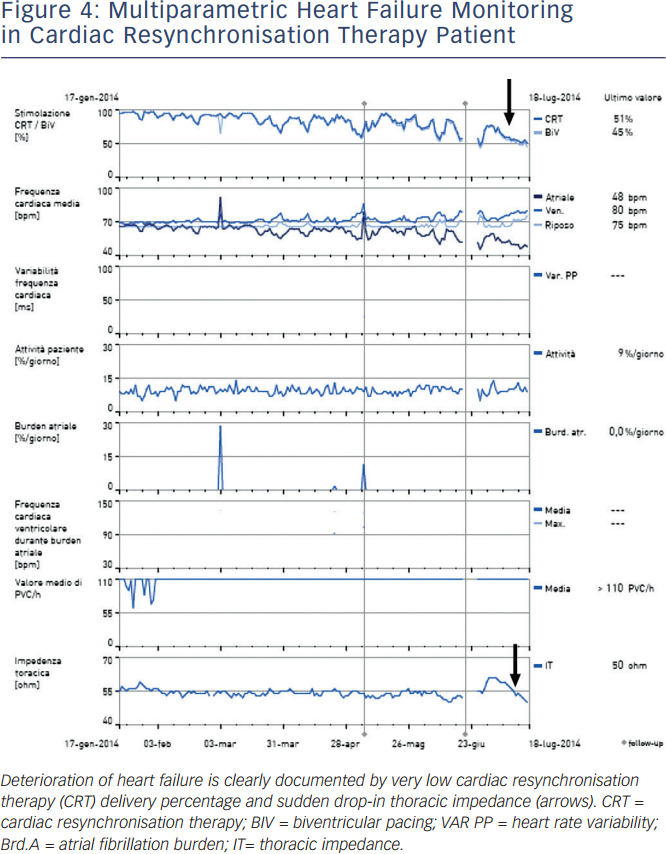
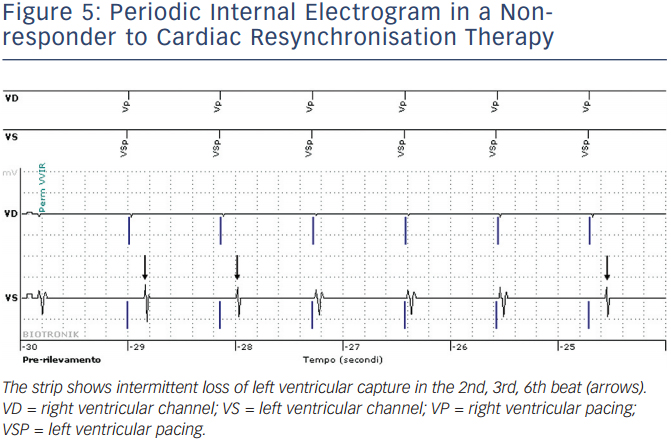
In addition to providing necessary therapies for cardiac arrhythmias and heart failure (HF), modern implantable devices also provide diagnostic information that may be useful in monitoring disease progression and in early detection of deterioration of HF, such as rest and night heart rate, heart rate variability, patient daily activity, percentage of right ventricular pacing in single and dual chamber devices, percentage of actual biventricular pacing in cardiac resynchronisation therapy (CRT) devices, intrathoracic impedance or hemodynamic sensors (see Figure 4). A periodic internal electrogram is available to check actual left ventricular capture (see Figure 5). A drop in impedance is related to pulmonary congestion and it may trigger an alert if it reaches a critical threshold. Continuous monitoring of device diagnostics may allow early identification of HF progression in the phase in which the patient is still asymptomatic, but in which filling pressures increase and sympathetic activation starts. The ultimate goal is to switch clinical reaction from a ‘reactive phase’, delivered when symptoms worsen and weight increases or when the patient has a pulmonary oedema, to a ‘proactive phase’ delivered when the patient is asymptomatic, typically two to three weeks in advance. The expected results of this strategy are prevention of hospitalisations for heart failure and of disease progression and improvement of patient quality of life (QoL). Algorithms based on impedance alone showed good sensitivity in HF early detection, on average two weeks in advance, but specificity was poor.29–32 The PARTNERS HF (Program to Access and Review Trending Information and Evaluate Correlation to Symptoms in Patients with Heart Failure) trial demonstrated that patients with a combined HF diagnostic algorithm had a 5.5-fold higher risk of HF event within 30 days.33 RM strategies have been associated with reduced unplanned device-related or cardiac in-hospital visits and reduced emergency visits for cardiac or device-related events (Evolution of Management Strategies of Heart Failure Patients with Implantable Defibrillators [EVOLVO] trial).34 Studies are ongoing to identify a combined score from device diagnostics with the greatest sensitivity and specificity for predicting HF events. Pressure-based technologies have been introduced to improve HF monitoring. Among them, a wireless implantable pulmonary artery haemodynamic monitoring system, the CardioMEMS™ (St Jude Medical, MN, US), has been recently approved by the US Food and Drug Administration (FDA). Pulmonary arterial pressure is continuously monitored and data may be reviewed by the physicians via a RM system. In the CardioMEMS Heart Sensor Allows Monitoring of Pressure to Improve Outcomes in NYHA Class III Heart Failure Patients (CHAMPION) trial35 (550 patients), patients randomised to CardioMEMS guided treatment had a 37 % significant reduction in HF hospitalisation when compared with the control group.
Survival
Evidence is emerging that RM may improve survival. The ALTITUDE database (a large observational retrospective, non-randomised, post-market analysis of real-world data) enrolled about 200,000 patients,36 of whom 30 % were remotely monitored. In the RM group risk of death was reduced by 50 % both in patients with single/ dual chamber ICDs and in those with CRT defibrillators (CRT-D). In the prospective randomised IN-TIME (The Influence of Implant- Based Home Monitoring on the Clinical Management of Heart Failure Patients with an Impaired Left Ventricular Function) trial,37 after one year patients in the RM arm, further to a significant improvement in the composite clinical Packer score (mortality plus HF hospitalisations plus New York Heart Association [NYHA] class), showed a 60 % reduction in cardiovascular mortality.
Implementing Remote Monitoring in Daily Practice – The Organisational Model
In spite of the documented benefits, implementing RM in daily clinical practice is challenging and is currently offered only to a minority of potential candidates.38 Reasons for that include reluctance to accept new technology, concern for legal issues, reimbursement issues, concern for increased work burden in the transition phase, and the need to develop new organisational models. Promising results have been demonstrated by a new model based on ‘Primary Nursing’ in which each patient is assigned to a nurse responsible for continuity of care.39The model is essentially based on a cooperative interaction between the roles of an expert reference nurse and a responsible physician with an agreed list of respective tasks and responsibilities. The model includes strict definition of workflow, early reaction, traceability, continuity of care and maintaining human relationship with the patient. Nurse duties primarily include patient training and education, website data entering, data and alert reviewing, data screening, critical case submission to the physician for clinical judgement, contacts with the patients, monitoring of patient compliance and therapy benefits. Written protocols are established in order to guide nurse reaction to findings and alerts. Physician duties include obtaining patient consent, analysis of submitted critical events and medical decisions, and communicating with general practitioners or other specialists. This model performed remarkably well in the wide Home Guide Registry which enrolled 1,650 patients.40,41 Sensitivity of RM in detecting major cardiovascular events was 84 % with a positive predictive value of 97%. RM required a median manpower less than one hour per health personnel per month for every 100 patients.
A centralised ‘hub and spoke’ model, in which one monitoring centre (hub) screened and filtered daily automatic data in pacemaker and ICD patients from several satellite clinics (spokes), has been suggested to help smaller centres fully utilise RM technology despite limited workforce and low patient numbers that may hamper development of dedicated, experienced, single-centre RM teams.42
The use of external centralised call centres has been suggested to reduce the work burden of the hospitals and to avoid the need for on-site dedicated expert teams.43 Personnel at the call centre usually include expert technicians on duty 24 hours a day, seven days a week, with electrophysiologists available on call. Potential advantages of this model may be represented by the 24-hour service and by the possibility of following thousands of patients in a centralised station. The main limits are represented by the loss of human relationship with the patient and potentially decreased patient compliance and satisfaction. Cost and effectiveness of this strategy could be unfavourable due to call centre costs and to the risk of repetitious alerts and duplication of clinical interventions.
Patient Acceptance and Satisfaction
Several studies have shown high patient satisfaction rates, ease of use and compliance with the use of RM systems, even when manual transmission of the data in non-wireless devices is requested.44–46 In spite of some initial concerns, RM has been demonstrated to be easy to use and well accepted even for elderly people and for patients with a low level of education.47 A few patients do not accept RM mainly due to concerns about technology and about the risk of losing the human contact with nurses and physicians. Patient education is critical to overcoming their concerns.48 Poor patient compliance may complicate workflow efficiency, mainly because of missed scheduled remote transmissions or duplicate transmissions. Phonecall burden due to patient noncompliance may negatively impact on personnel work load.49 Automaticity and reliability of the remote technology used is important. In the TRUST trial, no patient assigned to RM crossed over during the study and 98 % elected to retain this follow-up mode on trial conclusion, indicating patient acceptance and confidence in follow-up with this technology.50
Legal Issues
RM changes the paradigm of face-to-face visits by gathering electronic data into a data repository that is remote from the health facility, yet readily accessed and shared with various healthcare providers involved in a patient’s care or for research or educational purposes. There are challenges to maintaining the privacy of patient health information and potential issues related to liability for RM-related services. Telemedicine is still a medical act and the patient has to be appropriately informed about the service and must sign an informed consent document. In order to ensure privacy, patients have to sign a ‘Declaration of Privacy Principles’, written according to the local privacy laws. Responsibility for the data management process lies with the device manufacturer and RM service provider (usually the same), hospitals, telecommunication technology and service providers, physicians and allied professionals and patients. In this scenario the patient is proactive and asked for their compliance with guidelines and physician’s prescriptions. Patient compliance is critical for a good result, and in some cases RM should be denied or withdrawn.
The relationship between the patients and the clinical staff is based on an atypical contract, reported in the informed consent document signed by the patient and the physician, in which general rules for patient RM are defined. In particular, scheduling for alert and transmission revision, including the maximum reaction time, and the hospital service level agreement have to be understood and signed by the patient. The patient has to be informed that currently RM is not an emergency system, but only a tool to improve device surveillance and patient management. Clear instructions have to be given for emergency management. The technology provider is asked to respect the privacy statement and local laws, and to support the hospital centre in all aspects relating to providing patients with a RM service. Hospital duties are mainly to define and make available the facilities needed for RM.
Costs and Reimbursement Issues
Economical analyses have consistently demonstrated CIED RM to be cost effective.8,10,12,51–55 Mechanisms involved in savings include reduction of in-hospital visits, reduction of follow-up duration and physician and nurse time, and a reduction in patient costs related to travelling and missed work. Further economic benefits may come from increased device longevity, reduction of hospitalisations and prevention of clinical adverse events such as stroke. In the US since 2006 Medicare and Medicaid defined reimbursement codes for remote follow-up of pacemakers and defibrillators. Reimbursement policies for RM by healthcare systems and insurances vary widely among different countries in Europe.56 This limits adoption. Some form of reimbursement does exist in Germany, UK, Netherlands, Norway, Sweden, Finland, Denmark and Portugal. Although actual fee delivery to hospitals, doctors and manufacturers differs significantly. The fee-for-service payment approach and disease management global budget are the most common systems applied. No reimbursement exists in Switzerland, Belgium, Greece, Baltic States, Italy and Austria. This situation is rapidly evolving and soon hopefully homogeneous reimbursement rules will be established in Europe.
Current Guidelines
Since 2006, the Heart Rhythm Society (HRS)2 recommended that CIED manufacturers developed and implemented wireless and RM technologies to early identify abnormal device behaviour and to reduce under-reporting of device malfunctions. In 2008, the HRS/ European Heart Rhythm Association (EHRA) Expert Consensus on the Monitoring of Cardiovascular Implantable Electronic Devices1 stated that the majority of in-person follow-up could be replaced by RM. The document suggested to maintain face-to-face visits at predischarge visits, at one-month follow-up and at least once a year. Detail on CIED RM management recommendations has been published by a joint committee of the International Society for Holter and Noninvasive Electrocardiology (ISHNE) and EHRA4 in 2012 and by some national societies.57–59 In the 2013 European Society of Cardiology Guidelines on cardiac pacing and cardiac resynchronisation therapy it was stated that “Device-based RM should be considered in order to provide earlier detection of clinical problems and technical issues” (Recommendation class IIa, level of evidence A).60
Clinical Perspective
- Remote monitoring is rapidly becoming the new standard of care for patients with cardiac implantable electronic devices. The challenge is to implement remote monitoring in daily practice.
- The key to the success is from one side to invest on human resource (continuous education of personnel and development of the organisational model) and from the other to use technology progress for better interconnectivity and integration of data in the hospital electronic systems.
- Remote reprogramming which is not currently available, but technically feasible, will require great attention to patient safety prior to implementation.
- A clear reimbursement policy is mandatory to dedicate appropriate resource for a remote monitoring service.