Novel oral anticoagulants (NOACs) represent new options for preventing stroke in patients with atrial fibrillation (AF), and have been approved for use in North America and Europe. They carry a 50 % lower risk of intracranial haemorrhage compared with warfarin, no clear interactions with food, fewer interactions with medications and no need for frequent laboratory monitoring and dose adjustments. Although they lack a specific reversal agent, their use is increasing in the western world, thus imposing upon the dentists the task of performing invasive procedures in this setting with a continually higher frequency.
Magnitude of the Problem
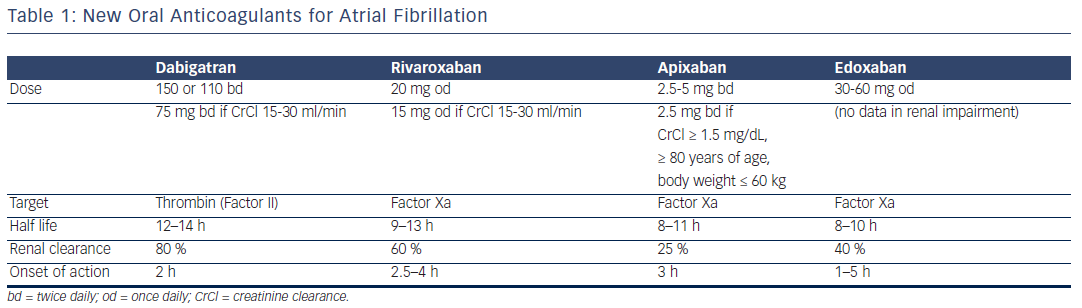
AF is the most common sustained arrhythmia in humans and affects 1–2 % of the general population worldwide. It affects three to six million people in the US,1,2 while in Asian countries its incidence is slightly lower.3,4 In the EU, 8.8 million adults over 55 years were estimated to have AF in 2010 and this number is expected to double by 2060 to 17.9 million.5 According to the first global assessment of AF, conducted within the framework of the Global Burden of Diseases (GBD), Injuries and Risk Factors Study, the estimated global prevalence of AF in 2010 was 33.5 million (20.9 million men and 12.6 million women), with almost five million new cases occurring each year.6 The prevalence of AF increases with age, from approximately 2 % in the general population, to 5–15 % at 80 years.2,7,8 Thus, AF represents a modern epidemic, and the practicing dentist is expected to deal with these patients at a continually increasing frequency.AF is associated with significant morbidity, including a two- to seven-fold increased risk for stroke (average 5 % per year).9–12 In the Framingham Study the percentage of strokes attributable to AF increases steeply from 1.5 % at 50–59 years of age to 23.5 % at 80–89 years of age.11 Approximately 20 % of all strokes are due to AF,13 and paroxysmal AF carries the same stroke risk as permanent or persistent AF.14 Thus, chronic anticoagulation is necessary for patients with AF and CHA2DS2VASc score ≥2, whereas no anticoagulation may only be recommended in patients with negligible risk and a score of 0. The novel oral anticoagulants are now recommended for nonvalvular AF as a potential alternative to warfarin by both the European Society of Cardiology (ESC) and American College of Cardiology (ACC)/American Heart Association (AHA) (see Table 1). Dabigatran is preferred to warfarin for non-valvular AF by the ESC13 and the Canadian Cardiology Society.15 NOACs are direct thrombin (dabigatran) or factor Xa (rivaroxaban, apixaban, edoxaban) inhibitors. They carry a 50 % lower risk of intracranial haemorrhage compared with warfarin, no clear interactions with food, fewer interactions with medications and no need for frequent laboratory monitoring and dose adjustments.16–18 Their main disadvantages are the lack of a reliable, specific antidote, specific assays to measure anticoagulant effect, and considerably higher cost than warfarin.19 NOACs do not interact with food but with inhibitors (or inducers) of P-glycoprotein transporters and cytochrome P450 (CYP) 3A4.A practical guide by the European Heart Rhythm Association (EHRA) has been published and a website created (www.NOACforAF.eu).20 The use of NOACs is continually increasing in the western world and, apart from AF, they are also used both for therapy and prevention of venous thromboembolism,21,22 i.e. pulmonary embolism and deep vein thrombosis, a disease entity with an annual incidence of approximately 1.2 cases per 1,000 adults.23,24
Assessment of Activity and Reversal of NOACs
Dabigatran
Activated partial thromboplastin time (aPTT) and thrombin clotting time (TCT or thrombin time) may be used for assessment of anticoagulant action, although not to guide dosage since the correlation is not linear. A normal aPTT indicates the absence of a significant dabigatran effect, whereas an aPTT >2.5 times the control 8–12 hours after dabigatran dosing is suggestive of excess anticoagulant activity.25 The Hemoclot® direct thrombin inhibitor assay (HYPHEN BioMed; France) provides an accurate measure of dabigatran drug levels.25 There is no specific antidote to dabigatran, but fresh frozen plasma and activated prothrombin complex concentrates (aPCC, 80 U/kg Factor Eight Inhibitor Bypassing Activity [FEIBA]; Baxter, Vienna, Austria) may be helpful.25–27 Recombinant activated factor VII (rVIIa, NovoSeven®, NovoNordisk, Bagsvaerd, Denmark) has also been proposed but data supporting its usefulness are lacking.28 Note that unlike the prothrombin complex concentrates (PCCs) in Europe and Canada, which contain all four vitamin K-dependent procoagulant proteins, those currently available in the US contain little or no factor VII.25
Apixaban and Rivaroxaban
Prothrombin time and, especially, anti-Xa assays (heparin) may be used as rough estimates of the anticoagulant effect. No specific antidotes exist, but the recombinant factor Xa andexanet alpha (Portola Pharmaceuticals; CA, USA) given as a bolus 600 or 720 mg and followed by an infusion of 4 mg/min for one hour, has been successfully tried. Infusion-related reactions and postural dizziness were the only side-effects seen in 10 % of patients.29 PCCs (50 IU/kg) are also recommended, and are probably preferred to aPCC.30 Apixaban and rivaroxaban are not removed by dialysis, being protein bound. Packed red cells in anaemia, platelet transfusions in patients receiving concurrent antiplatelet therapies, and fresh frozen in the presence of dilutional coagulopathy or disseminated intravascular coagulation, may also be tried as general measures.28
Edoxaban and betrixaban are Xa antagonists that have also been successfully tried in patients with non-valvular AF, but few data on their clinical use exist.
Dental Procedures in Patients on NOACs
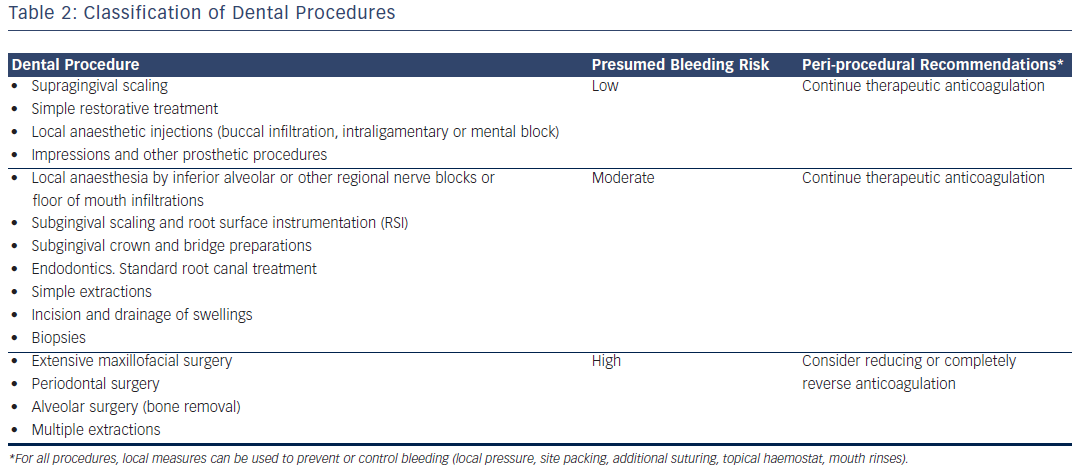
Dental treatment performed in patients receiving oral anticoagulant drug therapy is becoming increasingly common in dental offices. Frequently raised questions concern the accompanying thromboembolic and bleeding risks of the various anticoagulation regimens relative to invasive dental procedures. Many dental procedures do not involve a significant risk of bleeding and therefore no special measures are required when treating patients who take an oral anticoagulant drug. However, there are procedures that carry a risk of significant bleeding and for which the dentist must consider the management of the patient in relation to their anticoagulant therapy (see Table 2).
Peri-procedural Anticoagulation
In patients on warfarin, procedures at low bleeding risk do not require interruption of anticoagulation, provided the international normalised ratio (INR) is <4.0, whereas moderate and high-risk procedures are performed with an INR<1.5.31 Heparin bridging is not necessary, apart from in patients with certain mechanical valves. In a recent metaanalysis, heparin bridging for invasive procedures and surgery in patients receiving vitamin K antagonists for AF, prosthetic heart valve or venous thromboembolism conferred a greater than five-fold increased risk for bleeding, whereas the risk of thromboembolic events was not significantly different between bridged and nonbridged patients.32
Few data exist for NOACs. These agents are not indicated for anticoagulation in patients with prosthetic heart valves.33 Dentists, therefore, will encounter them in patients with AF and venous thromboembolism. There has been recent evidence that continuation or short-interruption of NOACs are safe strategies for most invasive procedures.34 Thus, dental procedures that involve manipulation of the gingival or periapical region of the teeth or perforation of the oral mucosa including uncomplicated teeth extractions do not require interruption of anticoagulation. For subgingival scaling, a small area should be scaled first to assess the amount of bleeding before instrumentation of larger areas is carried out. Local anaesthetics solutions containing a vasoconstrictor should be used unless contraindicated on other medical grounds. An aspirating syringe must be used for all local anaesthetic injections.35,36
Procedures with a relatively higher risk of bleeding such as multiple extractions or minor oral/maxillofacial surgery can also be safely performed without interruption of NOACs, provided they are carried out 12 hours after last dosing of dabigatran and 10 hours after last dosing of rivaroxaban or apixaban (see Table 1). Current available information suggests that the risk of bleeding in patients undergoing invasive dental procedures (for example up to three dental extractions, up to three dental implants and periodontal surgery) is low, provided that local haemostatic measures (suturing, ideally with resorbable sutures, gelatin sponge, gauze soaked in 5 % tranexamic acid, tranexamic acid mouth rinse) are used (see Table 3).37
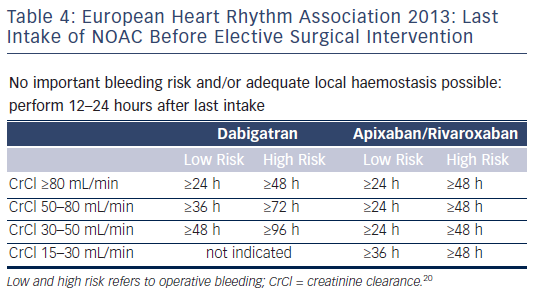
For more serious surgical operations, (major oral/maxillofacial surgery), the lack of data necessitates an empirical approach. In general, for surgical operations, depending on the risk of bleeding and renal function, preoperative interruption of NOACs for one to seven days has been recommended.25,38 The 2013 EHRA report recommends shorter intervals.20 Dabigatran should be discontinued for at least 24 hours (or longer in renal impairment) in patients requiring major oral/maxillofacial surgery (see Table 3).39 Additionally, consideration should be given to performing a thrombing clotting time (TT) or an aPTT 6–12 hours prior to surgery. The renal function of the patient should be also taken into account (see Table 4).
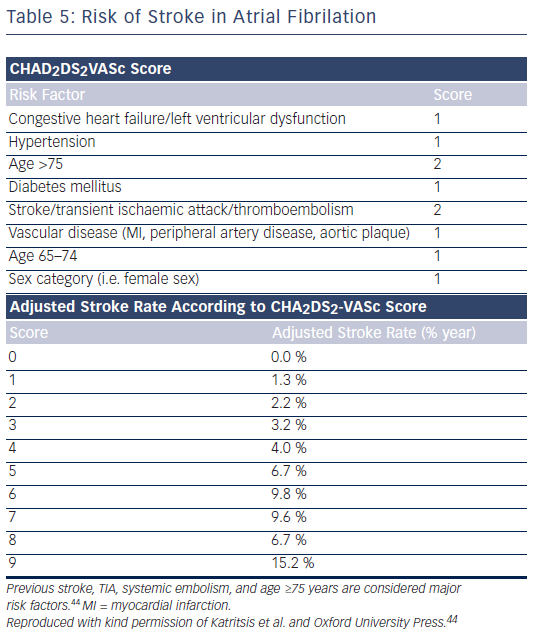
If discontinuation of anticoagulation is not considered safe and extensive oral surgery is required, peri-operative bridging anticoagulation with an appropriate dose of low molecular weight heparin (LMWH) or unfractionated heparin is recommended.39 It should be noted, however, that bridging with heparin is, on most occasions, not necessary and may increase the risk of bleeding.34 In a recent analysis of data from the Randomized Evaluation of Long-Term Anticoagulation Therapy (RELY) trial, interruption of dabigatran (two days) or warfarin (five days) for allowance of surgery was not associated by a significant occurrence of stroke and systemic embolism although heparin bridging was used in <80 % of patients on dabigatran, and major bleeding was not different in the two treatment groups.40 However, discontinuation of rivaroxaban in the ROCKET AF for at least three days was associated with a higher incidence of stroke compared with discontinuation of warfarin.41 Thus, in patients with a CHADS2DS2VASC score >4, i.e. >5 % annual risk of stroke (see Table 5), or those with a history of stroke who require temporary interruption of oral anticoagulation, bridging therapy with LMWH should be considered, especially when a newer oral anticoagulant is used.42 If urgent surgery or intervention is required in these patients, the risk of bleeding must be weighed against the clinical need for the procedure. Evaluation of common coagulation tests (aPTT for dabigatran; sensitive prothrombin time [PT] for FXa inhibitors) or of specific coagulation test (direct thrombin time [dTT] for dabigatran; chromogenic assays for FXa inhibitors) can be considered for assessment of anticoagulation intensity, but no clinical experience exists.20 Surgery should be deferred, if possible, until at least 12 hours and ideally 24 hours after the last dose. Nonspecific anti-haemorrhagic agents, such as rVIIa or PCCs, should not be given for prophylactic reversal due to their uncertain benefit: risk ratio.43 Reinitiation of these agents should be delayed for 24–48 hours and once a stable clot or complete haemostasis is assured, since within one to two hours of reinitiation the patient will be anticoagulated. For procedures with immediate and complete haemostasis NOACs can be resumed six to eight hours after the intervention.
Conclusions
Data on patients taking NOACs and who are undergoing dental procedures are scarce, and an empirical approach is inevitable regarding the management of these patients. Based on available evidence, the following recommendations can be made:
- In patients with normal renal function taking dabigatran, rivaroxaban or apixaban, simple invasive dental procedures can be carried out without interruption of the medication.
- All procedures should be performed as late as possible after the most recent dose, ideally >12 hours).
- Local haemostatic measures should be used routinely in these patients.
- Patients requiring oral/maxillofacial surgery may need discontinuation of oral anticoagulants for at least 24 hours pre-operatively, but always in consultation with treating physician.
- If stopped pre-operatively, NOACs should only be recommenced when a stable clot or adequate haemostasis has been achieved (typically 24–48 hours post-operatively).
- If post-operative bleeding occurs, oral anticoagulant therapy should be stopped, and local haemostatic measures applied.